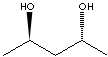
| (2R,4R)-(-)-PENTANEDIOL | ||
|
PRODUCT IDENTIFICATION |
||
| CAS NO. | 42075-32-1 |
|
| EINECS NO. |
|
|
| FORMULA | C5H12O2 | |
| MOL WT. | 104.15 | |
|
H.S. CODE |
||
| TOXICITY |
|
|
| SYNONYMS | (R,R)-(-)-2,4-Pentanediol; | |
|
DERIVATION |
|
|
|
CLASSIFICATION |
||
|
PHYSICAL AND CHEMICAL PROPERTIES |
||
| PHYSICAL STATE |
white crystals |
|
| MELTING POINT | 48 - 52 C | |
| BOILING POINT | 111 - 113 at 19 mm Hg | |
| SPECIFIC GRAVITY | ||
| SOLUBILITY IN WATER | ||
| pH | ||
| VAPOR DENSITY |
|
|
|
AUTOIGNITION |
||
|
NFPA RATINGS |
||
|
REFRACTIVE INDEX |
||
| FLASH POINT |
101 C |
|
| STABILITY |
Stable under ordinary conditions. Hygroscopic. |
|
|
GENERAL DESCRIPTION |
||
Chiral amino alcohols and their N-protected derivatives (Cbz, t-Boc, and Fmoc)
are versatile building blocks, auxiliaries, ligand or resolving
agent in asymmetric synthesis for biological research and pharmaceutical
applications such as antitumor, anesthetic, antipasmodic, hepatotoxic,
antiinflammatory or anti-HIV activities. They are used in the synthesis of
beta-blockers, HIV protease and ACE inhibitors, chiral auxiliaries and catalytic
enantioselective ligands. Their derivatives include the forms of;
|
||
| SALES SPECIFICATION | ||
|
APPEARANCE |
white crystals |
|
|
CHEMICAL PURITY |
99.5% min | |
|
OPTICAL PUIRTY |
99.5% min e.e. | |
| TRANSPORTATION | ||
| PACKING |
|
|
| HAZARD CLASS | ||
| UN NO. | ||
| OTHER INFORMATION | ||
| Hazard Symbols: XN, Risk Phrases: 16-36/37/38,
Safety Phrases: 22-24/25 Optical Rotation: -39° ~ -41° (C=10 in CHCl3) |
||