| ALLAPININ | |||||
|
PRODUCT IDENTIFICATION |
|||||
| CAS NO. | 97792-45-5 |
|
|||
| EINECS NO. | |||||
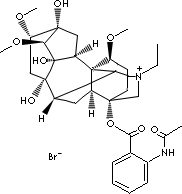
| FORMULA | C32H44N2O8.HBr | ||||
| MOL WT. | 665.61 | ||||
|
H.S. CODE |
|||||
| PRICE |
|
||||
| CLASSIFICATION |
|
||||
DERIVATION |
|
||||
| SYNONYMS | Allapinine; Lappaconiti hydrobromidum; | ||||
| Lappaconitine hydrobromide; (1alpha,14alpha,16beta)-20-Ethyl-1,14,16- trimethyoxyaconitane- 4,8,9-triol 4-(2-(acetylamino)benzoate), monohydrobromide; | |||||
|
PHYSICAL / CHEMICAL PROPERTIES |
|||||
| PHYSICAL STATE | white powder | ||||
| MELTING POINT |
|
||||
| BOILING POINT | |||||
| SPECIFIC GRAVITY | |||||
| SOLUBILITY IN WATER | Soluble | ||||
| pH | |||||
| VAPOR DENSITY |
|
||||
|
AUTOIGNITION |
|||||
|
NFPA RATINGS |
|||||
|
REFRACTIVE INDEX |
|
||||
| FLASH POINT |
|
||||
| STABILITY |
Stable under ordinary conditions. |
||||
|
EXTERNAL LINKS & GENERAL DESCRIPTION |
|||||
|
Aconitine, an alkaloid obtained from the plant Aconitum napellus is recognized for its phytomedical effects on the heart, central nervous system, and skeletal muscle (Catterall, Honerjager and Meissner, Ameri ). The arrhythmogenic effects of aconitine include the induction of premature ventricular contractions (PVC), ventricular tachycardia (VT), torsades de pointes, ventricular fibrillation (VF), and mortality in a dose-dependent manner (Lu and Clerck). Experimentally, aconitine-induced arrhythmias are thought to be induced by triggered activity due to delayed afterdepolarization and early afterdepolarization. At the molecular level, aconitine binds to Na+ channels and prolongs their open state favoring entry of a large quantity of Na+ into cytosol, which may be accompanied by Ca2+ overload via an electrogenic Na+-Ca2+ exchange (NCX) system and eventually induces triggered activity (Sawanobori et al., Watano et al.). Thus, the role of NCX is suspected to be important to generate triggered activity in the heart (Adaniya et al.,Sawanobori et al.). (source: http://jpet.aspetjournals.org/) The analgesic activity of lappaconitine, which is contained in the root of Aconitum sinomantanum Nakai, was examined after oral and subcutaneous administration to mice or rats by using methods for screening of analgesics, i.e., hot plate, tail immersion, tail pinch, tail pressure, acetic acid-induced writhing, bradykinin-induced flexor reflex of hind limb and Randall-Selitto methods. The results were compared with those for morphine, indometacin and acetylsalicylic acid (ASA). Analgesic activities of lappaconitine were greater than those of indometacin and ASA, but generally about 2 to 5 times less than those of morphine. However, in the rat tail immersion test, orally administered lappaconitine exhibited more potent analgesic activity than morphine; in this test, lappaconitine was almost equipotent when given orally and subcutaneously, whereas the potency of orally administered morphine was only one-twentieth of that of subcutaneously administered morphine. Like morphine, lappaconitine increased the pain threshold of the normal paw as well as that of the inflamed paw when tested by the Randall-Selitto method. The results show that lappaconitine has strong analgesic activity, and further suggest that the central nervous system may be involved in the action on the pain threshold. ( http://grande.nal.usda.gov/)The roots from Aconitum sp. plants have long been used in Chinese herbal medicine for treating pain and various heart conditions. The principal component of Aconitum remedies is usually aconitine, a site 2 neurotoxin that may induce severe neurological symptoms and cardiovascular collapse. Some Aconitum species also contain lappaconitine, the structure of which is remarkably similar to that of aconitine. In contrast to aconitine, a sodium channel agonist, lappaconitine reportedly blocks voltage-gated sodium channels in heart tissue. The results in the present study demonstrate that lappaconitine blocks cloned human heart (hH1) sodium channels under whole-cell, voltage-clamp conditions. Lappaconitine binding has several characteristics in common with the binding of site 2 neurotoxins, such as aconitine and batrachotoxin. For example, lappaconitine binds almost exclusively to open channels, but has little affect on resting or inactivated channels. Moreover, lappaconitine binding is inhibited by bupivacaine, a tertiary amine local anesthetic. Whereas site 2 neurotoxins often irreversibly modify channel kinetics, lappaconitine irreversibly blocks the channels. Finally, channels containing lysine substitutions within the local anesthetic receptor region at residues F1760 or N1765 are resistant to block by bupivacaine or lappaconitine. Given that site 2 neurotoxins and local anesthetics have nonidentical but overlapping binding regions, these data suggest that lappaconitine irreversibly blocks hH1 channels by binding to the site 2 receptor. ( http://molpharm.aspetjournals.org/)
|
|||||
| SALES SPECIFICATION | |||||
|
APPEARANCE |
white powder | ||||
|
ASSAY |
98.0% min | ||||
| TRANSPORTATION INFORMATION | |||||
| PACKING |
|
||||
| HAZARD CLASS | |||||
| UN NO. | |||||
| OTHER INFORMATION | |||||