| 2-NITRO-P-TOLUIDINE | ||
|
PRODUCT IDENTIFICATION |
||
| CAS NO. | 89-62-3 |
|
| EINECS NO. | 201-924-9 | |
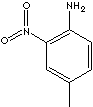
| FORMULA | CH3C6H3(NO2)NH2 | |
| MOL WT. | 152.15 | |
| H.S. CODE | ||
|
TOXICITY |
|
|
| SYNONYMS | 4-Methyl-2-nitroaniline; 4-Methyl-2-nitrobenzenamine; | |
| 3-Nitro-4-aminotoluene; 4-Amino-3-nitrotoluene; C.I. 37110; C.I. Azoic Diazo Component No. 8; Fast Red GL base; | ||
| DERIVATION |
|
|
|
CLASSIFICATION |
|
|
|
PHYSICAL AND CHEMICAL PROPERTIES |
||
| PHYSICAL STATE | orange powder | |
| MELTING POINT | 115 - 116 C | |
| BOILING POINT | 210 C (Decomposes) | |
| SPECIFIC GRAVITY | ||
| SOLUBILITY IN WATER |
|
|
| AUTOIGNITION |
|
|
| VISCOSITY | ||
| PH |
|
|
| VAPOR DENSITY | ||
| NFPA RATINGS | Health: 2; Flammability: 0; Reactivity: 4 | |
| FLASH POINT | ||
| STABILITY | Stable under ordinary conditions | |
|
GENERAL DESCRIPTION & APPLICATIONS |
||
|
Toluidine is the trivial name of methylaniline or aminotoluene. There are three
structural isomers, ortho-TOLUIDINE,
meta-TOLUIDINE, and para-TOLUIDINE. The
names of the three compounds indicate amino group positions relative to the
methyl group on the benzene ring. They can be obtained from coal tar as
by-products in the fractional distillation or can be prepared by reducing
nitrotoluene which are made by nitration of toluene. Toluidine is a compound
homologous with aniline (the simplest aromatic amine) and similar properties
aromatic amine. Toluidine is not soluble in water but in acidica queous
solution. o-Toluidine and m-Toluidine are clear to pale yellow, oily liquid.
But p-Toluidine solidifies to an off-white crystalline solid at about 43 C as it
has symmetrical structure . They are sensitive to air and light and tend to
darken on storage. They are toxic if inhaled, ingested, or absorbed through the
skin. Typical end-use markets for the toluidines and their derivatives are main
materials for the synthesis of imaging products like pigment, dyestuffs,and
photographic chemicals. All of these compounds are also used in the production
of antioxidants, agricultural, pharmaceutical and rubber chemicals.
The prefix nitro- indicates the presence of NO2- radical, while nitrate refers to any salt or ester of nitric acid or the NO3- anion. Nitroso- is the prefix indicating presence of the group -NO and azo- is for -N=N- group. Some range of organic compounds containing nitrogen include nitro compounds (RNO2 ), nitroso compounds (RNO), amines (R3N ), amino acids, and natural alkaloids or nucleotides. The nitrogen ion in nitro compounds is trigonally planar with 120° angles. There are two resonance bonds so that the two oxygens are equivalent. Nitro compounds are strongly basic due to electron withdrawing both inductively and mesomerically. Historically, they are abundant in dyes and explosives. Nitro compounds, organic hydrocarbons having one or more NO2 groups bonded via nitrogen to the carbon framework, are versatile intermediate in organic synthesis.
2-Nitro-p-toluidine is used as the base material for the synthesis colorants particularly pigment yellow 1 (Fast Yellow G) and red 3 (Toluidine red). |
||
| SALES SPECIFICATION | ||
|
APPEARANCE |
orange powder | |
| ASSAY |
98.0% min |
|
| MELTING POINT |
114 - 117 C |
|
| TRANSPORTATION | ||
| PACKING | 25kgs in bag | |
| HAZARD CLASS | 6.1 (Packing Group: III) | |
| UN NO. | 2660 | |
| OTHER INFORMATION | ||
|
Hazard Symbols: T N, Risk Phrases: 23/24/25-33-51/53, Safety Phrases: 36/37-45-61 |
||