| MANDELONITRILE | ||
|
PRODUCT IDENTIFICATION |
||
| CAS NO. | 532-28-5 |
|
| EINECS NO. | 208-532-7 | |
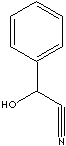
| FORMULA | C6H5CH(OH)CN | |
| MOL WT. | 133.15 | |
| H.S. CODE | ||
|
TOXICITY |
||
| SYNONYMS | alpha-Cyanobenzyl alcohol; Amygdalonitrile; | |
| alpha-Hydroxybenzeneacetonitrile; alpha-Hydroxyphenylacetonitrile; Benzaldehyde cyanohydrin; Hydroxyphenylacetonitrile; Mandelic acid nitrile; Phenylglycolonitrile; | ||
|
DERIVATION |
|
|
|
CLASSIFICATION |
|
|
|
PHYSICAL AND CHEMICAL PROPERTIES |
||
| PHYSICAL STATE | yellow to brown liquid | |
| MELTING POINT | -10 C | |
| BOILING POINT |
170 C (Decomposes) | |
| SPECIFIC GRAVITY |
1.117 | |
| SOLUBILITY IN WATER | Decomposes | |
|
SOLVENT SOLUBILITY |
||
| pH |
| |
| VAPOR DENSITY | ||
|
AUTOIGNITION |
| |
|
REFRACTIVE INDEX |
1.5305 - 1.5335 | |
|
NFPA RATINGS |
Health: 2; Flammability: 1; Instability: 0 | |
| FLASH POINT |
97 C | |
| STABILITY | Stable under ordinary conditions. Moisture sensitive. | |
|
DESCRIPTION AND APPLICATIONS |
||
|
Nitrile is an
organic compounds containing cyano group (-C��N, containing trivalent nitrogen)
which is attached to one carbon atom with the general formula RC��N. Their names
are corresponding to carboxylic acids by changing '-ic acid' to the suffix,
'-onitrile' which denotes only the ��N atom (triply bound) excluding the carbon
atom attached to it, or the suffix, '-carbonitrile' where the carbon atom in
the -CN is included, whichever preserves a single letter O. Examples are
acetonitrile from acetic acid and benzonitrile from benzoic acid. The prefix,
'cyano-' is used as an alternative naming system to indicate the presence of a
nitrile group in a molecule for the compounds of salts and organic derivatives
of hydrogen cyanide (HC��N). Isocyanides are salts and hydrocarbyl derivatives
from the isomer, HN+��C-. Sodium cyanide,
NaCN; potassium cyanide, KCN; calcium cyanide, Ca(CN)2; and hydrocyanic (or
prussic) acid, HCN are examples. Chemically, the simple inorganic cyanides
resemble chlorides in many ways. Organic nitriles act as solvents and are
reacted further for various application including;
·
Extraction solvent for fatty acids, oils and unsaturated hydrocarbons Mandelic Acid, phenylglycollic acid, is an alpha-hydroxy acid (AHA) which has a hydroxyl group on the carbon atom next to the acid group. If the hydroxy group is on the second carbon next to the acid group, it is called beta-hydroxy acid. Glycolic acid is the simplest AHA which has dual functionality of alcohol and acid in a low mole weight structure. Because of its small molecular weight and size, it has a better capability to penetrate skin. AHA is used extensively in cosmetics. It is known that it diminishes the lines on the skin and make skins look young by acting as a humectant to absorb moisture in air and by exfoliating action to break the bonds between dead skin cells. Mandelic Acid ( alpha-hydroxybenzeneacetic acid) is the smallest AHA among compounds which have aromatic group. It has an asymmetric carbon atom and thus has two chiral isomers; the dextro-, levo-. The D- and L-mandelic acid are enantiomers (also called enantiomorph; each molecule is asymmetrical and has the mirror image of the other) affect pharmaceutical activity. It is a white crystalline compound; melting at 118 C; partially soluble in water; freely soluble in isopropyl and ethyl alcohol; darkening upon exposure to light. Its structure provide the bacteriostatic property. It is excreted well in the urine. It is used as a antiseptic ingredient particularly against urinary tract infections. Mandelic acid and its derivatives are used to apply the dual activities as an antibacterial agent and as an antiaging agent (AHA activity) similar to glycolic acid. It is used as an intermediate for the synthesis of target molecules for other applications. Naturally occurring mandelic acid is found when amygdalin (a cyanogenetic glycoside found in many plants including bitter almond, apricot, and wild cherry) is spirit by hydrolysis with hydrochloric acid, while amygdalin is broken down into glucose, benzaldehyde, and prussic acid (hydrogen cyanide) in the presence of sulfuric acid. |
||
| SALES SPECIFICATION | ||
|
APPEARANCE |
yellow to brown liquid | |
|
ASSAY (G.C) |
95.0% min | |
|
INDIVIDUAL IMPURITY |
1.0% max | |
| TRANSPORTATION | ||
| PACKING | ||
| HAZARD CLASS | 6.1 (Packing group: I) | |
| UN NO. | 2810 | |
| OTHER INFORMATION | ||
| Hazard Symbols: T, Risk Phrases: 23/24/25, Safety Phrases: 24/25-45 | ||