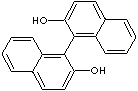
| beta-BINAPHTHOL | ||||
|
PRODUCT IDENTIFICATION |
||||
| CAS NO. | 602-09-5; 41024-90-2 |
|
||
| EINECS NO. | 210-014-0 | |||
| FORMULA | HOC10H6C10H6OH | |||
| MOL WT. | 286.32 | |||
|
H.S. CODE |
||||
| TOXICITY | ||||
| SYNONYMS | 2,2'-Dihydroxy-1,1'-dinaphthyl; 2,2'-Dihydroxybinaphthalene; | |||
| 1,1'-Bi-2-naphthol; 1,1'-Bis-2-naphthol; 2,2'-Dihydroxy-1,1'-binaphthalene; BINOL; 2,2'-Dihydroxy-1,1'-binaphthyl; 2,2'-Dihydroxydinaphthyl; 2,2'-Dinaphthol; Bis-beta-naphthol; Chiral binaphthol; alpha-Binaphthyl-2,2'-diol; (1,1'-Binaphthalene)-2,2'-diol; 1,1'-Binaphthyl-2,2'-diol; (±)-1,1'-Binaphthalene-2,2'-diol; (±)-2,2'-Dihydroxy-1,1'-dinaphthyl; | ||||
| PRICE | U$65/kg (50kgs), U$1,100.- (5kgs) | |||
|
CLASSIFICATION |
|
|||
|
PHYSICAL AND CHEMICAL PROPERTIES |
||||
| PHYSICAL STATE | white crystalline powder | |||
| MELTING POINT | 215 - 217 C | |||
| BOILING POINT | ||||
| SPECIFIC GRAVITY | ||||
| SOLUBILITY IN WATER | ||||
| pH | ||||
| VAPOR DENSITY | ||||
|
AUTOIGNITION |
|
|||
|
NFPA RATINGS |
Health: 2; Flammability: 1; Instability: 0 | |||
|
REFRACTIVE INDEX |
||||
| FLASH POINT | ||||
| STABILITY | stable under ordinary conditions. | |||
|
GENERAL DESCRIPTION AND APPLICATIONS |
||||
Naphthols are
either of two crystalline monohydric alcohols, derived from naphthalene and
belong to the phenol family. There are position isomers; alpha-naphthol (also
called 1-naphthol) is 1-hydroxynaphthalene and beta-naphthol (also called
2-naphthol) is 2-hydroxynaphthalene. The compound 1-naphthol is made by heating
1-naphthalenesulfonic acid with caustic alkali or by heating 1-naphthylamine
with water under pressure. The compound 2-naphthol is manufactured by fusing
2-naphthalenesulfonic acid with caustic soda. The different positions provide
various chemical structures which offer important roles to each characteristic
colours. Naphthols are slightly soluble in water but readily soluble in short
alcohols, ethers chlorinated solvents, and caustic alkalis. They are used
directly in making several dyes and are converted into numerous corresponding
amines, esters, ethers and carboxylic derivatives as well as into numerous
sulfo- and nitro-group substituted (mono-, di- and tri) naphthol compounds. They
find extensive applications in making dyes, pigments, fluorescent whiteners,
tanning agents, antioxidants, and antiseptics. Naphthol structure is found as a
ligand in transition-metal catalysts particularly in the form of binaphthol
(binol) which is composed of two naphthol rings connected at one carbon site on
each ring. Optically active binol is widely used in asymmetric synthesis of
rearrangements, epoxidations, reductions.
Wikipedia Linking:
http://en.wikipedia.org/wiki/Binaphthol
Chiral and Achiral Contributions to Sum-Frequency Generation from Optically Active Solutions of Binaphthol....The nonlinear sum- and difference-frequency generation spectroscopies can be probes of molecular chirality in optically active systems. We present a tensorial analysis of the chirality-specific electric-dipolar sumfrequency-generation susceptibility and the achiral electric-quadrupolar and magnetic-dipolar nonlinearities at second order in isotropic media. The chiral and achiral contributions to the sum-frequency signal from the bulk of optically active solutions of 1,1'-bi-2-naphthol (2,2'-dehydroxy-1,1'-binaphthyl) can be distinguished, and the former dominates. Ab initio computations reveal the dramatic resonance enhancement that the isotropic component of the electric-dipolar three-wave mixing hyperpolarizability experiences.....(http://people.ccmr.cornell.edu/) |
||||
| SALES SPECIFICATION | ||||
|
APPEARANCE |
white crystalline powder | |||
|
PURITY |
99.0% min |
|||
|
ALPHA-FORM |
Nil |
|||
| MELTING POINT | 215 - 217 C | |||
|
LOSS ON DRYING |
0.5% max |
|||
|
TRANSPORTATION |
||||
| PACKING | ||||
| HAZARD CLASS | 6.1 (Packing Group: III) | |||
| UN NO. | 2811 | |||
| OTHER INFORMATION | ||||
| Hazard Symbols: T, Risk Phrases: 25-36, Safety Phrases: 26-45 | ||||