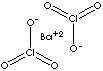| BARIUM CHLORATE | ||
|
PRODUCT IDENTIFICATION |
||
| CAS NO. | 13477-00-4
(Anhydrous), 10294-38-9 (Monohydrate) |
|
| EINECS NO. | 236-760-7 | |
| FORMULA | Ba(ClO3)2 | |
| MOL WT. | 304.23 | |
|
H.S. CODE |
||
| TOXICITY | ||
| SYNONYMS | Chloric Acid, Barium Salt; | |
| SMILES | ||
|
CLASSIFICATION |
|
|
|
PHYSICAL AND CHEMICAL PROPERTIES |
||
| PHYSICAL STATE | white crystalline solid | |
| MELTING POINT |
|
|
| BOILING POINT | ||
| SPECIFIC GRAVITY | 3.18 | |
| SOLUBILITY IN WATER | soluble (27.5g/ml) | |
| pH | ||
| VAPOR DENSITY | ||
|
AUTOIGNITION |
||
|
NFPA RATINGS |
Health: 1 Flammability: 0 Reactivity: 1 Other: Oxidizer | |
|
REFRACTIVE INDEX |
|
|
| FLASH POINT | Not combustible | |
| STABILITY | Stable under ordinary conditions. | |
|
APPLICATIONS |
||
| Barium Chlorate is used in the pyrotechnics to give a bright light for illumination (especially green light). It is a powerful oxidizing agent. It is used to manufacture explosives and matches because of its ability to produce oxygen. Chlorate is a powerful ingredient of bleaching powder used in paper and pulp processing and calico printing. | ||
| SALES SPECIFICATION (MONOHYDRATE) | ||
|
APPEARANCE |
white crystalline solid | |
| ASSAY |
98.0%
min
|
|
|
PARTICLE SIZE |
1.0% max (bigger than 150µm) |
|
|
Fe |
0.003% max |
|
|
WATER |
5.8% max |
|
| TRANSPORTATION | ||
| PACKING | 100kgs in drum | |
| HAZARD CLASS | 5.1 (Packing Group: II) | |
| UN NO. | 1485 | |
| OTHER INFORMATION | ||
| Hazard Symbols: XN O, Risk Phrases: 9-20/22, Safety Phrases:13-27 | ||
| Chlorate is the common name for chlorate(V) salt. The related names to the acids are clear when the oxidation number statement is appended; hypochlorite [or chlorate(I)] contains the ion, ClO- ; chlorite [or chlorate(III)], ClO2-; Chlorate [or chlorate(V)], ClO3-; perchlorate [or chlorate(VII)], ClO4-. | ||