| CHLOROSULFONIC ACID | ||
|
PRODUCT IDENTIFICATION |
||
| CAS NO. | 7790-94-5 |
|
| EINECS NO. | 232-234-6 | |
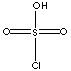
| FORMULA | ClSO2OH | |
| MOL WT. | 116.52 | |
| H.S. CODE | 2806.20 | |
| TOXICITY |
| |
| SYNONYMS | Monochlorosulfuric acid; CSA; Acide chlorosulfonique; | |
| Sulfuric Chlorohydrin; Sulfonic Acid, Monochloride; 氯磺酸; | ||
| SMILES | sulfur anhydride | |
|
CLASSIFICATION |
|
|
|
PHYSICAL AND CHEMICAL PROPERTIES |
||
| PHYSICAL STATE | clear to light yellow fuming liquid | |
| MELTING POINT |
-80 C | |
| BOILING POINT |
151 - 152 C | |
| SPECIFIC GRAVITY |
1.753 | |
| SOLUBILITY IN WATER | Decomposes (to sulfuric acid and hydrochloric acid) | |
| pH | ||
| VAPOR DENSITY | 4 | |
|
AUTOIGNITION |
| |
|
NFPA RATINGS |
Health: 4; Flammability: 0; Reactivity: 2 | |
|
REFRACTIVE INDEX |
| |
| FLASH POINT |
| |
| STABILITY | Stable under ordinary conditions. | |
|
APPLICATIONS |
||
|
Sulfonic acid is a compound with general formula RSO2OH, where R is an aliphatic
or aromatic hydrocarbon. It is a derivative of sulfuric acid (HOSO2OH) where an
OH has been replaced by a carbon group or a compound where a hydrogen atom has
been replaced by treatment with sulfuric acid; for example, benzene is converted
to benzenesulfonic acid (water-soluble). Sulfonic acid has a sulfur atom bonded
to a carbon atom of a hydrocarbon and bonded also to three oxygen atoms, one of
which has been attached to a hydrogen atom. Sulfonic acid is acidic due to the
hydrogen atom, stronger than a carboxylic acid. Sulfonic acid is one of the most
important organo sulfur compounds in organic synthesis. Sulfonic acids are used
as catalysts in esterification, alkylation and condensation reactions.
Sulfonates are salts or esters of sulfonic acid. Sulfonic salts are soluble in
water. Sulfonic acid and its salts present in organic dyes provide useful
function of water solubility and or improve the washfastness of dyes due to
their capabiltity of binding more tightly to the fabric. They are widely used in
the detergent industry. Alkylbenzene sulfonic acid is the largest-volume
synthetic surfactant because of its relatively low cost, good performance, the
fact that it can be dried to a stable powder and the biodegradable environmental
friendliness. Sulfonate cleaners do not form an insoluble precipitates in hard
water. Sulfonic acid salts and esters are intermediates widely used in organic
synthesis and particularly phenolic compounds and cation exchange resins. They
are synthetic intermediate for a number of biologically active compounds and
pharmaceutical candidates such as sulfa drugs. Short carbon chain metallic
sulfonate is used in electroplating which is a process for applying a metallic
coating on a metal surface by electrodeposition from a suitable electrolyte
solution for imparting corrosion resistance and direct production of printed
circuit boards without etching out of a piece of copper sheet.
Chlorosulfonic Acid is a corrosive, clear liquid; melting point -80 C; boiling point 151 C; soluble in chlorinated solvents. It is hygroscopic and lachrymatory strongly. It reacts with water to form HCl. Chlorosulfonic Acid is used to produce organic sulfonic aicds for the end applications of detergent, pharmaceuticals, pesticides, and dyes. It was a starting material to produce saccharin. |
||
| SALES SPECIFICATION | ||
|
APPEARANCE |
clear to light yellow liquid | |
| CONTENT | 98.0% min | |
| SULFURIC ACID |
2.0% max | |
| Fe |
0.01% max | |
| TRANSPORTATION | ||
| PACKING | Iso-Tank | |
| HAZARD CLASS | 8 (9.2) | |
| UN NO. | 1754 | |
| OTHER INFORMATION | ||
| Hazard Symbols: T+ C, Risk Phrases: 14/35/37, Safety Phrases: 26/45 | ||