|
NITROTOLUENES
| ||||||||||||||||||||||||||||||
|
GENERAL | ||||||||||||||||||||||||||||||
|
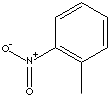
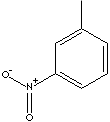
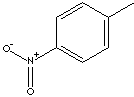
ortho-NITROTOLUENE, meta-NITROTOLUENE, and para-NITROTOLUENE. The three
compounds are structural isomers. Nitrotoluenes are produced commercially, as a
mixture, by nitration of toluene. Typical end-use markets for the Nitrotoluenes
and their derivatives are main materials for the synthesis of imaging products
like pigment, dyestuffs,and photographic chemicals. All of these isomers are
also used in the production of antioxidants, agricultural, pharmaceutical and
rubber chemicals.
The prefix nitro- indicates the presence of NO2- radical, while nitrate refers to any salt or ester of nitric acid or the NO3- anion. Nitroso- is the prefix indicating presence of the group -NO and azo- is for -N=N- group. Some range of organic compounds containing nitrogen include nitro compounds (RNO2 ), nitroso compounds (RNO), amines (R3N ), amino acids, and natural alkaloids or nucleotides. The nitrogen ion in nitro compounds is trigonally planar with 120° angles. There are two resonance bonds so that the two oxygens are equivalent. Nitro compounds are strongly basic due to electron withdrawing both inductively and mesomerically. Historically, they are abundant in dyes and explosives. Nitro compounds, organic hydrocarbons having one or more NO2 groups bonded via nitrogen to the carbon framework, are versatile intermediate in organic synthesis.
Nitro compounds are readily reduced into amines when reacted with hydrochloric acid. Aromatic nitro compounds also yield anilines, aromatic amine compounds. Nitro compounds are cleaved into two parts by the addition of a molecule of water to produce carbonyl compounds (aldehydes or ketones). Aromatic nitro compounds undergo nucleophilic substitutions to be replaced by the hydroxide anion, resulting in the creation of phenol compounds and by alkoxy nucleophiles to corresponding ethers. | ||||||||||||||||||||||||||||||
|
ortho-NITROTOLUENE |
meta-NITROTOLUENE |
para-NITROTOLUENE | ||||||||||||||||||||||||||||
|
|
|
| ||||||||||||||||||||||||||||
|
CAS
No. :88-72-2 |
CAS
No. : 99-08-1 |
CAS
No. : 99-99-0 | ||||||||||||||||||||||||||||
|
APPLICATION |
||||||||||||||||||||||||||||||
|
o-NITROTOLUENE |
||||||||||||||||||||||||||||||
|
SPECIFICATION |
||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||
|
SYNONYMS |
||||||||||||||||||||||||||||||
|
o-NITROTOLUENE | ||||||||||||||||||||||||||||||