|
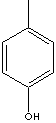
para-CRESOL |
||
|
PRODUCT IDENTIFICATION |
||
| CAS NO. |
106-44-5 |
|
| EINECS NO. | 203-398-6 | |
| FORMULA | CH3C6H4OH | |
| MOL WT. | 108.14 | |
| H.S. CODE | 2907.12.0000 | |
|
TOXICITY |
Oral rat LD50: 207 mg/kg | |
| SYNONYMS | p-Cresol; p-Methylphenol; p-Methylphenylol; | |
| p-Hydroxytoluene; p-Oxytoluene; p-Toluol; 1-Hydroxy-4-methylbenzene; 4-Hydroxytoluene; 4-Methylphenol; p-Cresylic acid; Paracresol; p-Kresol (German); 4-Cresol; | ||
|
SMILES |
c1(ccc(O)cc1)C | |
|
CLASSIFICATION |
|
|
|
EXTRA NOTES |
|
|
|
PHYSICAL AND CHEMICAL PROPERTIES |
||
| PHYSICAL STATE | Clear to slightly amber semi solid, phenolic odor | |
| MELTING POINT | 33 - 34 C | |
| BOILING POINT |
195 - 205 C |
|
| SPECIFIC GRAVITY | 1.02 - 1.03 | |
| SOLUBILITY IN WATER | 23 g/l | |
|
SOLVENT SOLUBILITY |
Soluble in alcohol, ether |
|
| pH | ||
| VAPOR DENSITY | 3.72 | |
| AUTOIGNITION |
550 C |
|
| NFPA RATINGS | Health: 3; Flammability: 2; Reactivity: 0 | |
|
REFRACTIVE INDEX |
||
| FLASH POINT | 86 - 87 C | |
| STABILITY | Stable under ordinary conditions | |
|
EXTERNAL LINKS & GENERAL DESCRIPTION |
||
|
Drug Information Portal (U.S. National Library of Medicine) - p-Cresol PubChem Compound Summary - p-Cresol KEGG (Kyoto Encyclopedia of Genes and Genomes) - p-Cresol http://www.ebi.ac.uk/ - p-Cresol http://www.ncbi.nlm.nih.gov/ - p-Cresol http://www.atsdr.cdc.gov/ http://oehha.ca.gov/ Local:
Cresols undergo electrophilic substitution reactions such as chlorination, bromination, sulfonation and nitration at the vacant position. They also undergo condensation reactions with aldehydes, ketones or dienes. O-cresol is a starting material for the synthesis of herbicides such as 4,6-dinitro-o-cresol (DNOC) and 2-methyl-4-chlorophenoxyacetic acid (MCPA). Meta-cresol is used in the manufacture of explosives. Meta and para-cresol are used in phenol-formaldehyde resins and are converted to tricresyl phosphate used as a plasticizer and gasoline additive and antioxidants such as di-tert-butylcresols (BHT). Ortho- and para-cresols are used in the production of lubricating oils and motor fuels. |
||
| SALES SPECIFICATION | ||
|
APPEARANCE |
Clear to slightly amber semi solid | |
|
PURITY |
98.0% min | |
|
ISOMERS |
1.8% max |
|
|
DISTILLATION RANGE |
189 - 192 C |
|
|
SULFUR |
Nil |
|
|
PHENOL |
0.5% max |
|
| SPECIFIC GRAVITY | 1.02 - 1.03 at 70 C | |
| MOISTURE |
0.2% max |
|
| TRANSPORTATION | ||
| PACKING | 200kg in drum | |
| HAZARD CLASS | 6.1 (Packing Group: II ) | |
| UN NO. |
3455 |
|
| SAFETY INFORMATION | ||
|
HAZARD OVERVIEW |
OSHA Hazards: Highly toxic by inhalation, Toxic by ingestion, Toxic by skin absorption. Target Organs: Central nervous system, Lungs, Eyes, Liver, Kidney |
|
|
GHS |
|
|
| SIGNAL WORD |
Danger |
|
|
PICTOGRAMS |
|
|
|
HAZARD STATEMENTS |
H301-H311-H314 |
|
|
P STATEMENTS |
P280-P301 + P310-P305 + P351 + P338-P310 |
|
| EC DIRECTIVES |
|
|
| HAZARD CODES |
|
|
|
RISK PHRASES |
24/25-34 |
|
|
SAFETY PHRASES |
36/37/39-45 |
|
| PRICE INFORMATION | ||
|
|
||