1,6-HEXANEDIOL
PRODUCT IDENTIFICATION
629-11-8, 140434-69-1
TOXICITY
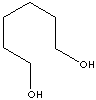
SMILES
CLASSIFICATION
Diol, Monomer
PHYSICAL AND CHEMICAL PROPERTIES
white waxy solid
SOLVENT SOLUBILITY
REFRACTIVE INDEX
101 C
EXTERNAL LINKS & GENERAL DESCRIPTION
http://www.inchem.org/
SHORT
SUMMARY WHICH SUPPORTS THE REASONS FOR THE CONCLUSIONS AND RECOMMENDATIONS.
The production volume of this chemical in Germany was 10,000-50,000t
in 1991. The total production volume is used as an intermediate
in chemical industry for the synthesis of polyesters and polyesterol-type
polyurethanes, which are used for paints, laquers and varnishes.
GENERAL DESCRIPTION: Diols contribute to high water solubility, hygroscopicity and reactivity with many organic compounds, on usually linear and aliphatic carbon chain. 1,6-Hexanediol, linear diol containing two primary hydroxyl groups at terminal locations for reaction of di-substitution, is used as an intermediate in polymer syntheses (nylon, polyesters manufacturing). The main application field is polyurethanes manufacturing. It is also used in gasoline refining and in pharmaceutical manufacturing. Alcohols are very weak acids as they lose H+ in the hydroxyl group. Alcohols undergoes dehydration reaction which means the elimination of water molecule replaced by a pi bond between two adjacent carbon atoms to form alkenes under heating in the presence of strong acids like hydrocloric acid or phosphoric acid. Primary and secondary alcohols can be oxidized to aldehydes and ketones respectively. Carboxylic acids are obtained from oxidation of aldehydes. Oxidation in organic chemistry can be considered to be the loss of hydrogen or gain of oxygen and reduction to gain hydrogen or loss of oxygen. Tertiary alcohols do not react to give oxidation products as they have no H attached to the alcohol carbon. Alcohols undergoes important reactions called nucleophilic substitution in which an electron donor replaces a leaving group, generally conjugate bases of strong acids, as a covalent substitute of some atom. One of important reaction of alcohol is condensation. Ethers are formed by the condensation of two alcohols by heating with sulfuric acid; the reaction is one of dehydration. Almost infinite esters are formed through condensation reaction called esterification between carboxylic acid and alcohol, which produces water. Alcohols are important solvents and chemical raw materials. Alcohols are intermediates for the production of target compounds, such as pharmaceuticals, veterinary medicines, plasticizers, surfactants, lubricants, ore floatation agents, pesticides, hydraulic fluids, and detergents.
APPEARANCE
white waxy solid
1,6-HEXANEDIOL
96.0% min
C6 DIOLS
99.0% min
ACID NUMBER
0.1 max (mg KOH/g)
COLOR, APHA
20 max
WATER
0.1% MAX
LINEAR DIOLS
|
Linear Diol |
CAS RN |
EINECS |
Melting point |
Boiling Point |
Formula |
Mole WT. |
|
Pentane-1,2-diol |
5343-92-0 | 226-285-3 |
|
206 C | CH3CH2CH2CH(OH)CH2OH | 104.15 |
|
Pentane-1,5-diol |
111-29-5 | 203-854-4 |
|
239 - 242 C | HO(CH2)5OH | 104.15 |
| Hexane-1,2-diol | 6920-22-5 | 230-029-6 |
|
223 - 224 C | CH3(CH2)3CH(OH)CH2OH | 118.17 |
| Hexane-1,6-diol | 629-11-8 | 211-074-0 | 38-42 C |
250 C |
HO(CH2)6OH | 118.17 |
| Heptane-1,2-doil | 3710-31-4 |
|
|
|
CH3(CH2)4CH(OH)CH2OH | 132.20 |
| Heptane-1,7-diol | 629-30-1 | 211-085-0 | 17-19 C | 259 C | HO(CH2)7OH | 132.20 |
| Octane-1,2-diol | 1117-86-8 | 214-254-7 | 36-38 C | 131 C at 10 mmHg | CH3(CH2)5CH(OH)CH2OH | 146.23 |
| Octane-1,8-diol | 629-41-4 | 211-090-8 | 58-61 C | 172 C at 20 mmHg | HO(CH2)8OH | 146.23 |
| Nonane-1,3-diol | 23433-07-0 | 245-660-2 |
|
|
CH3(CH2)5CH(OH)C2H5OH | 160.26 |
| Noname-1,9-diol | 3937-56-2 | 223-517-5 | 45-47 C | 288 C | HO(CH2)9OH | 160.26 |
| Decane-1,2-diol | 1119-86-4 | 214-288-2 | 48-50 C |
255 C |
CH3(CH2)7CH(OH)CH2OH | 174.28 |
| Decane-1,10-diol | 112-47-0 | 203-975-2 |
74 C |
|
HO(CH2)10OH | 174.28 |
| Undecane-1,2-diol | 13006-29-6 | 235-848-2 |
|
|
CH3(CH2)8CH(OH)CH2OH | 188.31 |
| Undecane-1,11-diol | 765-04-8 | 212-135-4 |
|
|
HO(CH2)11OH | 188.31 |
| Dodecane-1,2-diol | 1119-87-5 | 214-289-8 | 56-60 C |
|
CH3(CH2)9CH(OH)CH2OH | 202.33 |
| Dodecane-1,12-diol | 5675-51-4 | 227-133-9 | 79-81 C |
324 C |
HO(CH2)12OH | 202.33 |
PRICE INFORMATION