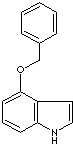
PRODUCT IDENTIFICATION
H.S. CODE
TOXICITY
CLASSIFICATION
EXTRA NOTES
PHYSICAL AND CHEMICAL PROPERTIES
57 - 61 C
REFRACTIVE INDEX
Stable under ordinary conditions.
EXTERNAL LINKS & GENERAL DESCRIPTION
Drug Information Portal (U.S. National Library of Medicine) - 4-Benzyloxyindole
PubChem Compound Summary - 4-Benzyloxyindole
http://www.ebi.ac.uk/chebi/ - 4-Benzyloxyindole
http://www.ncbi.nlm.nih.gov/ - 4-Benzyloxyindole
http://www.sigmaaldrich.com/
Application:
Reactant for regioselective synthesis of oxopyrrolidine analogs
via iodine-catalyzed Markovnikov addition ·Reactant for preparation
of indoles by Bartoli reductive cyclization as useful intermediates
in medicinal chemistry research ·Reactant for synthesis of
carbon-11-labeled 4-aryl-4H-chromenes as new PET agents for imaging
of apoptosis in cancer·Reactant for preparation of HCV inhibitors·Reactant
for preparation of indol-3-yl tetramethylcyclopropyl ketones as
CB2 cannabinoid receptor ligands·Reactant for preparation
of 4-aryl-4H-chromenes as apoptosis inducers
Local:
Indole,
benzopyrrole, is a yellow crystalline powder with unpleasant aroma. It has the
pyrrole ring (five-membered unsaturated ring structure composed of four carbon
atoms and one nitrogen atom) which is fused to benzene ring. There are
tautomerer called indolenine (unsubstituted 3H-indole) and structural isomer,
isoindole. But they are unstable. Indole occurs in some plants or in coal tar,
and is formed in the intestine during putrefaction and by certain cultures of
bacteria. it is commercially synthesized from phenylhydrazine and pyruvic acid.
Indole structure is a motif in nature. Prominent
examples include tryptophan (aromatic side chain amino acid), serotonin
(neurotransmitter), auxin (plant growth hormone), and indigo (plant colorant).
One more interesting point is all these compounds have functional branches at 3
position. Indole is used in perfumery and in preparing tryptophan,
one of the 20 amino acids commonly found in animal proteins. It has important
application in the industry of plant growth. It is used to prepare indoleacetic
acid (auxin) and other plant growth substances which help the development of
roots in plant. It is used to make selective herbicides. Indole and its
derivatives are widely used in making perfumes, dyes and agrochemicals as well
as in biologically
active ingredients and clinical diagnostics.
APPEARANCE
ASSAY
99.0% min
MELTING POINT
57 - 61 C
HAZARD OVERVIEW
OSHA Hazards: Irritant
GHS
PICTOGRAMS

HAZARD STATEMENTS
H315-H319-H335
P STATEMENTS
P261-P305 + P351 + P338
![]()
RISK PHRASES
36/37/38
SAFETY PHRASES
26-36