|
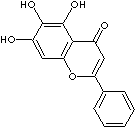
BAICALEIN |
| Synonyms. Baicalein; 5,6,7-Trihydroxy-2-phenyl-4H-1-benzopyran-4-one; 5,6,7-Trihydroxyflavone; 5,6,7-trihydroxy-2-phenyl-chromen-4-one; 5,6,7-trihydroxy-2-phenylchromen-4-one; Noroxylin; Noroxylin; 5,7-Trihydroxyflavone; |
|
|
| PRODUCT IDENTIFICATION | |
|
CAS RN |
491-67-8 |
|
EINECS RN |
|
|
FORMULA |
C15H10O5 |
|
MOLE WEIGHT |
270.24 |
|
H.S CODE |
2933.99.7900 |
|
SMILES |
c1(cc(c2c(o1)cc(c(c2O)O)O)=O)c1ccccc1 |
|
CLASSIFICATION |
Flavone, Antioxidant, Hormone antagonist, Prostaglandin antagonist |
|
EXTRA NOTES |
A trihydroxyflavone with the hydroxy groups at positions C-5, -6 and -7.(ChEBI) |
|
|
| PHYSICAL AND CHEMICAL PROPERTIES | |
|
PHYSICAL STATE. |
white to pale-yellow solid |
|
MELTING POINT |
264 - 265 C |
|
BOILING POINT |
|
|
DENSITY |
|
|
SOLUBILITY IN WATER |
Insoluble |
| SOLVENT SOLUBILITY | Soluble in alcohol, methanol, ether, acetone, ethyl acetate, hot glacial acetic acid. Sparingly soluble in Chloroform |
|
VAPOR DENSITY |
|
|
log P(octanol-water) |
3.27 |
|
VAPOR PRESSURE |
|
|
AUTOIGNITION TEMP |
|
| pH |
|
|
REFRACTIVE INDEX |
|
|
FLASH POINT |
|
|
|
| STABILITY AND REACTIVITY | |
| STABILITY | Stable under normal conditions. |
|
INCOMPATIBLE MATERIALS |
Strong oxidizing agents, Strong bases, moisture |
| POLYMERIZATION |
Has not been reported |
|
NFPA RATINGS |
Health: 1, Flammability:0, Reactivity: 0 |
|
|
| EXTERNAL LINKS & GENERAL DESCRIPTION | |
|
Wikipedia Linking - Baicalein Google Scholar Search - Baicalein Drug Information Portal (U.S. National Library of Medicine) - Baicalein PubChem Compound Summary - Baicalein KEGG (Kyoto Encyclopedia of Genes and Genomes) - Baicalein http://www.ebi.ac.uk/ - Baicalein http://www.ncbi.nlm.nih.gov/ - Baicalein http://ci.nii.ac.jp/
|
|
|
| SALES SPECIFICATION |
|
|
APPEARANCE |
white to pale-yellow solid |
|
CONTENT |
98% min (HPLC) |
|
PARTICLE SIZE |
80 mesh (100%) |
| RESIDUE ON IGNITION |
1.0% max |
|
RESIDUAL SOLVENTS |
EU2000 |
|
HEAVY METALS |
20ppm max |
|
ARSENIC |
2ppm max |
|
LOSS ON DRYING |
3.0% max |
| MICROBIOLOGY |
Aerobic Plate Count: 100 organisms/g max |
|
|
| TRANSPORT & REGULATORY INFORMATION | |
|
UN NO. |
Not regulated |
| HAZARD CLASS |
|
| PACKING GROUP | |
|
|
| SAFETY INFORMATION | |
|
HAZARD OVERVIEW |
Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
|
GHS |
|
|
SIGNAL WORD |
Warning |
|
PICTOGRAMS |
|
|
HAZARD STATEMENTS |
H315-H319-P335 |
|
P STATEMENTS |
P261-P305 + P351 + P338 |
| EC DIRECTIVES |
|
| HAZARD CODES |
|
|
RISK PHRASES |
36/37/38 |
|
SAFETY PHRASES |
26-36 |
|
|
| PACKING |
|
Preserve in light-resistant and well-closed containers |
|
|