SPIRONOLACTONE
PRODUCT IDENTIFICATION
H.S. CODE
TOXICITY
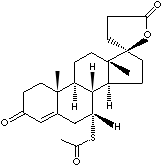
[C@@]12([C@@]3([C@@H](CC1)[C@H]1[C@H](CC3)[C@@]3(C(= CC(=O)C C3) C[C@H]1SC(=O)C)C)C)OC(=O)CC2
CLASSIFICATION
Aldosterone antagonist, Diuretic, Expectorant, Hormone Antagonist, Natriuretic
EXTRA NOTES
Overall Carcinogenic Evaluation: Group 3
PHYSICAL AND CHEMICAL PROPERTIES
22 mg/l
AUTOIGNITION
NFPA RATINGS
REFRACTIVE INDEX
Stable under ordinary conditions
EXTERNAL LINKS & GENERAL DESCRIPTION
Wikipedia Linking - Spironolactone
Google Scholar Search - Spironolactone
Drug Information Portal (U.S. National Library of Medicine) - Spironolactone
PubChem Compound Summary - Spironolactone
Drug Bank - Spironolactone
KEGG (Kyoto Encyclopedia of Genes and Genomes) - Spironolactone
http://www.ebi.ac.uk/ - Spironolactone
http://www.ncbi.nlm.nih.gov/ - Spironolactone
http://toxnet.nlm.nih.gov/
Hazardous
Substances Data Bank - Spironolactone
Local:
Spironolactone is a is a synthetic 17-lactone steroid belong to a class of drugs
called potassium-sparing diuretics which inhibit the exchange of sodium for
potassium and hydrogen ions in the distal tubule. It acts as a competitive
aldosterone antagonist in the distal renal tubules. Anti-androgen effect is also
utilized as a component of hormone replacement therapy for transgender, in the
treatment of hair loss, female hirsutism and acne but it can lead to adverse
effects. Spironolactone, as a potassium-sparing diuretic, increases the
secretion of water and sodium, while decreasing the excretion of potassium.
Aldosterone synthesis is stimulated by increased plasma angiotensin II or
potassium levels, which are present in proportion to plasma sodium deficiencies.
Spironolactone is mainly used in the treatment of refractory edema in patients
with congestive heart failure, nephrotic syndrome, or hypokalemia.
Spironolactone itself is a weak diuretic, and it is used in combination with
other diuretics. Angiotensin II antagonist such as valsartan, eprosartan and
telmisartan decrease the effects of aldosterone and used as antihypertensive.
Members of aldosterone antagonists include;
|
Aldosterone Antagonist |
CAS RN |
| 17alpha-Hydroxy-6beta,7beta-methylene-3-oxo-D-homo-17aalpha-pregna-4,16- dien-21- carboxylic acid | 81844-75-9 |
| 17beta-Hydroxy-12-alpha-etiojerv-4-en-3-one | 22785-18-8 |
| 17-Hydroxy-3-oxo-17alpha-pregn-4-ene-7alpha,21-dicarboxylic acid, 7-(1-methylethyl) ester potassium | 41020-87-5 |
| 17-Hydroxy-3-oxo-19-nor-17alpha-pregn-4-ene-21-carboxylic acid gamma-lactone | 1722-54-9 |
| 18-Deoxyaldosterone | 2507-88-2 |
| 3,20-Dioxopregn-4-ene-18'-carboxaldehyde cyclic 18'-(1,2-ethandiylmercaptal) | 123376-04-5 |
| 5-Oxo-A-tetranor-17-alpha-pregna-9,11-diene-21-17-carbolactone | 76225-44-0 |
| 6,7-Epoxycanrenone | 114577-01-4 |
| 7alpha-(Methylthio)spironolactone S-oxide | 38753-75-2 |
| 7alpha-Thiospironolactone | 38753-76-3 |
| 7-Thiomethylspirolactone | 38753-77-4 |
| Aldatense | 76270-05-8 |
| Canrenoate Potassium | 2181-04-6 |
| Canrenone | 976-71-6 |
| Dicirenone | 41020-79-5 |
| Dihydroxy-6,7-dihydrocanrenone | 67483-35-6 |
| Drospirenone | 67392-87-4 |
| Eplerenone | 107724-20-9 |
| Mespirenone | 87952-98-5 |
| Mexrenoate potassium anhydrous | 41020-67-1 |
| Mexrenoate potassium | 43169-54-6 |
| Mexrenone | 41020-65-9 |
| Oxprenoate potassium | 76676-34-1 |
| Prorenoate | 49848-01-3 |
| Prorenoate potassium | 49847-97-4 |
| Prorenone | 49848-04-6 |
| Spironolactone | 52-01-7 |
| Spirorenone | 74220-07-8 |
APPEARANCE
IDENTIFICATION
Pass Test A, B, C
ASSAY
OPTICAL ROTATION
MELTING POINT
134 - 136 C
LOSS ON DRYING
0.5% max
CHLORIDE
50ppm max
SULFATED ASH
0.1% max
1.0% max
IMPURITY
1.0% max
HAZARD OVERVIEW
Harmful. May be harmful if swallowed. This substance has caused adverse reproductive and fetal effects in animals. May cause eye and skin irritation. May cause respiratory and digestive tract irritation. This substance has caused reproductive effects in humans. This product contains spironolactone, a substance known to the state of California to cause cancer. Target Organs:Kidney, Male reproductive system., Female reproductive system., Bone marrow, Skin
GHS
PICTOGRAMS


HAZARD STATEMENTS
H360
P STATEMENTS
P201-P308 + P313
![]()
RISK PHRASES
60
SAFETY PHRASES
53-22-36/37/39-45