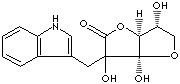
ASCORBIGEN
PRODUCT IDENTIFICATION
H.S. CODE
TOXICITY
CLASSIFICATION
EXTRA NOTES
PHYSICAL AND CHEMICAL PROPERTIES
off-white to yellow powder
AUTOIGNITION
NFPA RATINGS
REFRACTIVE INDEX
Stable under ordinary conditions. Moisture sensitive.
EXTERNAL LINKS & GENERAL DESCRIPTION
Drug Information Portal (U.S. National Library of Medicine) - Ascorbigen
PubChem Compound Summary - Ascorbigen
Drug Bank - Ascorbigen
KEGG (Kyoto Encyclopedia of Genes and Genomes) - Ascorbigen
http://www.ebi.ac.uk/chebi/ - Ascorbigen
http://www.ncbi.nlm.nih.gov/ - Ascorbigen
http://linkinghub.elsevier.com/retrieve/
Ascorbigen (ABG) belongs to the glucosinolate family and occurs mainly in
Brassica vegetables. It is formed by its precursor glucobrassicin.
Glucobrassicin is enzymatically hydrolyzed to indole-3-carbinol, which in turn
reacts with l-ascorbic acid to ABG. The degradation of glucobrassicin is induced
by plant tissue disruption. The ABG formation depends on pH and temperature. The
degradation of ABG in acidic medium causes a release of l-ascorbic acid and a
formation of methylideneindolenine; in more alkaline medium, the degradation of
ABG causes the formation of 1-deoxy-1-(3-indolyl)-��-l-sorbopyranose and
1-deoxy-1-(3-indolyl)-��-l-tagatopyranose. ABG may partly mediate the known
anticarcinogenic effect of diets rich in Brassicacae. Furthermore, ABG is able
to induce phase I and II enzymes that are centrally involved in the
detoxification of xenobiotics. Cosmeceuticals containing ABG as an active
principle are becoming increasingly popular, although the underlying cellular
and molecular mechanisms regarding its potential antiaging and
ultraviolet-protective properties have not been fully established.
http://www.ncbi.nlm.nih.gov/
Ascorbigen (ABG) belongs to the glucosinolate
family and occurs mainly in Brassica vegetables. It is formed by its precursor
glucobrassicin. Glucobrassicin is enzymatically hydrolyzed to indole-3-carbinol,
which in turn reacts with L-ascorbic acid to ABG. The degradation of
glucobrassicin is induced by plant tissue disruption. The ABG formation depends
on pH and temperature. The degradation of ABG in acidic medium causes a release
of l-ascorbic acid and a formation of methylideneindolenine; in more alkaline
medium, the degradation of ABG causes the formation of
1-deoxy-1-(3-indolyl)-alpha-L-sorbopyranose and
1-deoxy-1-(3-indolyl)-alpha-l-tagatopyranose. ABG may partly mediate the known
anticarcinogenic effect of diets rich in Brassicacae. Furthermore, ABG is able
to induce phase I and II enzymes that are centrally involved in the
detoxification of xenobiotics. Cosmeceuticals containing ABG as an active
principle are becoming increasingly popular, although the underlying cellular
and molecular mechanisms regarding its potential antiaging and
ultraviolet-protective properties have not been fully established.
http://www.ncbi.nlm.nih.gov/
New derivatives of
2-C-/(indolyl-3)methyl/-beta-L-threo-L-glycero-3- hexulofuranosono -1,4-lactone,
vis. 1'-ethylascorbigen, 1'-benzylascorbigen and
1',2'-dimethyl-5'-methoxyascorbigen were obtained. In aquous solutions at
physiological temperature and pH values ascorbigens disintegrate rather rapidly
to yield L-ascorbic acid, the rate of the latter's formation depending on the
substituent in the indole nucleus and increasing with growth of pH and
temperature. Intraperitoneal injection of 1'-methylascorbigen to mice leads to
steady rise of L-ascorbic acid level in blood plasma.
Local
Ascorbigens:
|
Product |
CAS RN |
|
Ascorbigen |
8075-98-7 |
| Ascorbigen A | 26676-89-1 |
| 1'-Methylascorbigen | 92557-61-4 |
| 1'-Ethylascorbigen | 110326-15-3 |
APPEARANCE
off-white to yellow powder
97.0% min (HPLC)
Hazard Overview
Not known
PRICE INFORMATION