PRODUCT IDENTIFICATION
208-288-1
H.S. CODE
TOXICITY
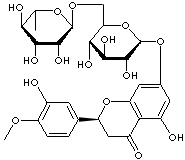
C1[C@@H](c2cc(c(OC)cc2)O)Oc2cc(O[C@@H]3O[C@H](CO[C@H]4[C@@H] ([C@@H]([C@@H](O)[C@@H](O4)C)O)O)[C@@H](O)[C@@H]([C@H]3O)O) cc(c2C1=O)O
CLASSIFICATION
Vitamin, Flavanone glycoside, Flavonoid, Bitter compound
EXTRA NOTES
A flavanone glycoside found in citrus fruit peels.
Biochem/physiol Actions: Flavonoid phytochemical extracted from citrus species. It is the rhamnoglucoside (rutinoside) of hesperetin.
PHYSICAL AND CHEMICAL PROPERTIES
AUTOIGNITION
NFPA RATINGS
REFRACTIVE INDEX
Stable under ordinary conditions. Hygroscopic, light sensitive.
EXTERNAL LINKS & GENERAL DESCRIPTION
Wikipedia Linking - Hesperidin
Google Scholar Search - Hesperidin
Drug Information Portal (U.S. National Library of Medicine) - Hesperidin
PubChem Compound Summary - Hesperidin
Drug Bank - Hesperidin
KEGG (Kyoto Encyclopedia of Genes and Genomes) - Hesperidin
http://www.ebi.ac.uk/ - Hesperidin
http://www.ncbi.nlm.nih.gov/ - Hesperidin
Local:
Hesperidin, a polyphenolic bioflavonoid, is a water-soluble pigment synthesized or naturally occurring as a glycoside in orange peel or other citrus fruits. It, chemically (S)-7-[[6-O-( 6-deoxy- alpha-L-mannopyranosyl )- beta-D-glucopyranosyl] oxy]- 2,3-dihydro-5-hydroxy- 2-( 3-hydroxy-4-methoxyphenyl )-4H-1-Benzopyran-4-one, is a white to yellow crystalline powder melting at ca 260 C. Bioflavonoids are called phytochemicals, Vitamin P, and Vitamin C2. There are about 2000 known
bioflavonoids including catechin, citrin, eriodictin, hesperetin, hesperidin,
nobiletin, quercetin, rutin, sinensetin, and tangeretin. Vitamin P is not a
dietary essential but is known to have the effect on capillary disorders. It
increases the strength of the walls of the blood capillaries and regulates their
permeability. It may have an contribution to the total antioxidant activity and
detoxifiers. It may enhance the action of Vitamin C. It is considered that it
lowers down cholesterol levels if it taken with Vitamin C. It is also know to
have pharmacological action as an anti-inflammatory ,antihistaminic and
antiviral agent. Hesperidin is the predominant flavonoid in lemons and sweet oranges.
Rutin, the glycoside form of quercetin, is a bioflavonol having aromatic trimeric heterocyclic structure. It is a naturally occurring pigment. It, chemically 3-[[6-O-(6-deoxy-alpha-L- mannopyranosyl )-beta-D-glucopyranosyl] oxy]-2- (3,4-dihydroxyphenyl) -5,7-dihydroxy-4H- 1-Benzopyran-4-one, is a yellow to greenish crystalline powder melting at 190 C. Rutin is classified as vitamin P which increases the strength of the walls of the blood capillaries and regulates their permeability. Others classified as vitamin P are catechin, citrin, eriodictin, hesperetin, hesperidin, nobiletin, quercetin, sinensetin, and tangeretin. It is not a dietary essential but is known to have the effect on capillary disorders. It may also have an contribution to the total antioxidant activity and detoxifiers. It is also know to have pharmacological action as an anti-inflammatory ,antihistaminic and antiviral agent.
Members of rutin molecules
|
Product |
CAS RN |
| Rutinose | 90-74-4 |
| Rutin | 153-18-4 |
| Hesperidin | 520-26-3 |
| Narcissin | 604-80-8 |
| Tetra(hydroxyethyl)rutoside | 6980-20-7 |
| Troxerutin | 7085-55-4 |
| Ascorutin | 8067-12-7 |
| Rutin sodium sulfate | 12768-44-4 |
| Eriocitrin | 13463-28-0 |
| Narirutin | 14259-46-2 |
| Tamarixetin 7-rutinoside | 14265-53-3 |
| Delphinidin-3-rutinoside | 15674-58-5 |
| Kaempferol 7-neohesperidoside | 17353-03-6 |
| Keracyanin | 18719-76-1 |
| Monoxerutin | 23869-24-1 |
| Veronicastroside | 25694-72-8 |
| Ethoxazorutoside | 30851-76-4 |
| Isorhamnetin-3-O-rutinoside-7-O-glucoside | 55481-91-9 |
| Rutin S | 64755-26-6 |
| Diethanolamine-rutin | 69344-84-9 |
| Rutin-N-mustard | 85625-48-5 |
| Nirurin | 96253-68-8 |
| Rutin sulfate | 108916-86-5 |
| Isorhamnetin-3-O-rutinoside-4'-O-glucoside | 111393-97-6 |
| Violdelphin | 126417-59-2 |
| Swertiamacroside | 128585-97-7 |
| D-Glucose-rutin | 130603-71-3 |
| Rutin butyryl ester | 131981-32-3 |
| 2',5,5',7-Tetrahydroxy-flavanone-7-O-rutinoside | 153286-43-2 |
| Rutin hydrate | 207671-50-9 |
|
Rutin trihydrate |
250249-75-3 |
APPEARANCE
ASSAY
Pass
-65° ~ -70° (C=2 Pyridine)
5.0% max
5.0% max
HEAVY METALS
10ppm max
GENERAL DESCRIPTION OF FLAVONOID
- Flavonols (Hydroxy derivatives of flavone): Fisetin, Galangin, Kaempferide, Kaempferol, Morin, Myricetin, Myricitrin, Quercetin, Quercetrin, Rhamnetin, Robinin, Rutin, Spirenoside
- Flavones (skeleton: 2-phenylchromen-4-one): Apigenin, Baicalein, Chrysin, Diosmetin, Diosmin, Flavone, Luteolin, Rpoifolin,Tangeretin, Techtochrysin, Rhamnazin, Nobiletin, Natsudaidain.
- Isoflavones (skeleton: 3-phenylchromen-4-one): Daidzin, Genistein, Irilone, Luteone, Prunetin, Pratensein,
- Flavonones (derivation by reduction of the 2(3) C=C bond): Eriodictyol, Hesperidin, Hesperetin, Likvirtin, Naringin; Naringenin; Pinocembrin
- Flavanols (derivation by reduction of the keto group):(+)-Catechin, (+)-Gallocatechin, (-)-Epicatechin (EC), (-)-Epigallocatechin (EGC), (-)-Epicatechin 3-gallate (ECG), (-)-Epigallocatechin 3-gallate (EGCG), Theaflavin, Theaflavin 3-gallate, Theaflavin 3'-gallate, Theaflavin 3,3' digallate, Thearubigins
- Anthocyanidins (aglycones of the glycoside anthocyanins): Apigeninidin, Cyanidin, Delphinidin, Diosmetinidin, Guibourtinidin, Fisetinidin, Luteolinidin, Malvidin, Pelargonidin, Peonidin, Robinetinidin, Tricetinidin, Capensinidin, Petunidin, Europinidin, Aurentinidin, Columnidin, 5-Desoxy-malvidin, 5-Desoxy-peonidin, Hirsutinidin, Rosinidin