|
CAS
NO.
|
17455-13-9 |
|
| EINECS NO. |
241-473-5 |
| FORMULA |
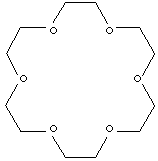
C12H24O6 |
| MOL
WT. |
264.32 |
|
H.S.
CODE
|
2932.99.0090 |
|
TOXICITY
|
Oral rat LD50;
525 mg/kg |
| SYNONYMS |
18-Crown 6-Ether;
|
1,4,7,10,13,16-Hexanoxacyclooctadecane;
|
Cyclic Hexamer
of Ethylene oxide; Crown ether
18-crown-6; Hexaoxacyclo-octadecane; Other
RN: 63172-42-9; 65154-22-5; 66037-87-4; 71210-94-1; 71211-03-5;
71245-01-7; 71251-42-8; 71251-39-3; 71251-38-2; 134316-24-8;
168081-58-1 |
|
SMILES
|
C1COCCOCCOCCOCCOCCO1 |
|
CLASSIFICATION
|
CROWN ETHERS
/ Host-Guest Chemistry |
|
PHYSICAL AND CHEMICAL PROPERTIES
|
| PHYSICAL
STATE |
white
crystals |
| MELTING POINT |
42
- 45 C |
| BOILING
POINT |
116
C |
| SPECIFIC GRAVITY |
1.111
- 1.113 |
| SOLUBILITY
IN WATER |
soluble
(7.49E+04 mg/l at 25 C) |
| pH |
|
| VAPOR DENSITY |
|
|
AUTOIGNITION
|
510
C
|
| pKa |
(Dissociation Constant
at 20 C) |
| log Pow |
-0.68
(Octanol-water) |
| VAPOR
PRESSURE |
6.67E-05 (mmHg at 25 C) |
|
HENRY'S LAW |
3.45E-13 (atm-m3/mole at 25 C) |
| OH RATE |
8.41E-11 (cm3/molecule-sec
at 25 C Atmospheric) |
|
NFPA
RATINGS
|
|
|
REFRACTIVE
INDEX
|
|
| FLASH
POINT |
112
C
|
| STABILITY |
Stable
under ordinary conditions. Moisture
sensitive |
|
GENERAL
DESCRIPTION & EXTERNAL LINKS
|
|
Crown Ether is a macrocyclic polyether whose structure contains hydrogen, carbon
and oxygen atoms. Each oxygen atoms are confined between two carbon atoms and
exhibits a conformation with a hole (accordingly called "crown"). The common
names of Crown Ethers have a prefix to designate the total number of atoms in
the cycle and a suffix to designate the number of oxygen atoms in the cycle. For
example, 15-crown-5 is composed of 15 atoms in the cycle, 5 of which are O and
10 of which are C. The main characteristic crown ethers is the complexation of
the ether oxygens with various ionic species. If metalic elements pass through
the center of the hole, they stick to oxygen atoms. The crown compound is then
termed "host-guest" chemistry; Crown Ether acts as the "host" taking ionic
species as its "guest". Crown compounds locks guest atoms in a solution and wrap
around it. Crown ethers are used in PTC (phase-transfer catalysts) system and in
encouraging the solubility of inorganic compounds in organic solvents to promote
chemical reaction. 'Host-Guest' chemistry of crown ethers can be a key to
identify the move of essential elements in the body and can play the part of
very complicated biological reactions such as enzyme's functions, which can be
applied to develop new pharmaceuticals. Wikipedia
Linking: http://en.wikipedia.org/wiki/Crown_ether
http://www.org-chem.org/
....An interesting thing about a crown ether is that the ring can capture a
positively charged ion like a metal ion and an ammonium ion (the sphere shown
inside the ring is the positively charged ion). The negatively charged electrons
of the oxygen atoms pointing inward are used to attract and catch the ion. This
property made some conventionally impossible things possible, including pulling
ionic substances into organic solvent. I should emphasize here the importance of
the ring structure, as it holds onto an ion ten thousand times more strongly
than does the similar open chain structure.
Larger ions fit in larger crown
ethers, and smaller ions fit in smaller crown ethers. The ionic radius increases
in the order of lithium, sodium, and potassium, and these metal ions are known
to be most compatible with twelve-, fifteen-, and eighteen-membered rings,
respectively.
It’s also known that two crown ether molecules can sandwich an
ion. In these cases, an ion larger than the natural fit can be captured. A crown
ether with an interesting “switch” property has been synthesized...........
|
| SALES
SPECIFICATION |
|
APPEARANCE
|
white
crystals |
|
ASSAY |
98.0%
min |
| TRANSPORTATION |
| PACKING |
|
| HAZARD CLASS |
|
| UN
NO. |
|
| OTHER
INFORMATION |
|
Hazard Symbols: XN,
Risk Phrases: 22, Safety
Phrases: |
|
GENERAL DESCRIPTION OF ETHER |
|
Ether is any of a number of organic compounds characterized by an oxygen atom
joined with single bonds by two carbon atoms that are part of hydrocarbon
groups. The general formula is R-O-R', where R and R' are alkyl or aromatic
groups. Ethers are formed by the condensation of two alcohols by heating with
sulfuric acid; the reaction is one of dehydration. Ethers can be prepared from
alkyl halide reacted with metallic alkoxide (called Williamson synthesis).
Ethers are similar to alcohols but are generally less dense, less soluble in
water, and have lower boiling points. They are relatively unreactive, which
makes them valuable solvents. But ethers will be
cleaved at high temperatures by concentrated hydrogen halides. Ethers have relatively low boiling point
compare to alkanes as they don't form hydrogen bonds each other. Ethers are more
lipophilic than esters [R-C(=O)-O-R']or amides [RCO-NH2]. Ethers are widely used as solvents for various organic reactions because they
are relatively the least reactive among common organic compounds except alkanes
and fluorocarbons. The common reaction of ethers is cleavage of the C–O bond by
strong acids either in linear chain or cyclic structure. Ethers in which oxygen
is bonded to primary and secondary alkyl groups can form peroxide compounds in
the presence of gaseous oxygen due to two unpaired electrons in oxygen. Ethers can act as
Lewis bases in chemical reactions. Commonly, ethers are
named simply in listing the alkyl groups in alphabetical order or alkane order
such as ethyl methyl ether or methyl ethyl ether, which is methoxyethane in
IUPAC nomenclature ( the formula of "alkoxyalkane" ). When ether is a
parts of complex molecule or aromatic derivatives, it is described as an alkoxy
substituent such as methoxybenzene ( trivial name is anisole). The methoxy
prefix indicates the function methyl group joined by single bonds to an oxygen atom,
with the general formula -O-CH3. Cyclic ethers
have ring structure where the oxygen has become part of the ring. The term of
epoxide indicate three membered cyclic ether (also called oxirane) in which an
oxygen atom is joined to each of two carbon atoms that are already bonded to
each other; four
membered cyclic ether is called oxetane; five membered cyclic ether, furan (or
oxolane); six membered cyclic ether, pyran (also called oxane) respectively.
Their unhindered oxygen atom carries two unshared pairs of electrons - a
structure which favors the formation of coordination complexes and the solvation
of cations. Cyclic ethers are used as important solvents, as chemical
intermediate and as monomer for ring-opening polymerization. Crown Ether is a macrocyclic polyether whose structure contains
hydrogen, carbon and oxygen atoms. Each oxygen atoms are confined between two
carbon atoms and exhibits a conformation with a hole (accordingly called
"crown"). Anisole is one of the simplest aromatic compound to
which ether group is linked. But
it is different with aromatic compounds like furan where the oxygen is a part of
the ring. Anisole, C6H5OCH3
(methyl phenyl ether), is a clear liquid that is soluble in ether and
alcohol; insoluble in water; boiling point 155 C. Anisole and its derivatives
are used as solvents and in perfumery. Anisole can be obtained from anise seed.
Anisic acid, p-methoxybenzoic acid, is a part of cresol class antiseptic
compounds. It
is also used as an insect repellent and ovicide.
Anisole, anisic acid, and their derivatives are also widely used in
chemical reaction as intermediates to obtain target materials such as dyes,
pharmaceuticals, perfumes, photoinitiators and agrochemicals. |
