| CAS
NO. |
3001-15-8 |
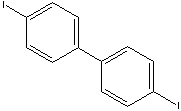
|
| EINECS
NO. |
221-080-5 |
| FORMULA |
C12H8I2 |
| MOL
WT. |
406.00 |
|
H.S.
CODE
|
|
|
TOXICITY
|
|
| SYNONYMS |
4,4'-Diiodobifenil
(Spanish); 4,4'-Diiodobiphényl (French); |
| SMILES |
|
|
CLASSIFICATION
|
|
|
PHYSICAL
AND CHEMICAL PROPERTIES
|
| PHYSICAL
STATE |
yellow crystals |
| MELTING
POINT |
201
- 204 C |
| BOILING
POINT |
|
| SPECIFIC
GRAVITY |
|
| SOLUBILITY
IN WATER |
|
| pH |
|
| VAPOR
DENSITY |
|
| AUTOIGNITION |
|
| NFPA
RATINGS |
|
|
REFRACTIVE
INDEX
|
|
| FLASH
POINT |
|
| STABILITY |
Stable
under ordinary conditions |
|
APPLICATIONS
|
Iodine is a
nonmetallic halogen element in Group 17 of periodic table; atomic number 53;
atomic mass 126.9; melting point ca 114 C; boiling point ca 184 C; specific
gravity 4.93 g/cm3; Oxidation states: 7,5,1,-1; [Kr] 4d10 5s2 5p5. Iodine is a
nearly black poisonous, corrosive solid at room temperature and readily sublimes
to a deep violet vapour, the colour of which is responsible for its name from
Greek. It is insoluble in water, soluble in common solvents. Iodine is required
in small amounts in human body for the function of the thyroid gland. Iodine
forms many important compounds of iodine such as iodine(V)oxide, potassium
iodide, iodine trichloride and iodoform of an Iodine containing organic
compound. Iodine is used as a component in germicides and disinfectants with
surfactants to carry iodine. Potassium iodide is used as a heat stabilizer and a
catalyst in synthetic fiber manufacturing. Organic iodides and
Iodine metal salts are used in formulating
antiseptics and x-ray contrast media compounds in pharmaceuticals. Potassium
iodate and other forms like calcium iodate and ethylene diamine dihydroiodide
are used in animal feeds to prevent deficiency of iodine. Iodine and its organic
compounds are useful in dye industry for the high bright colors as well as
pharmaceutical industry. An example of organic Iodine, iodoform has weak
bactericidal qualities and is used to treat minor skin
diseases. |
| SALES
SPECIFICATION |
|
APPEARANCE
|
yellow crystals |
|
PURITY
|
99.0%
min
|
|
WATER
|
0.15%
max
|
| TRANSPORTATION |
| PACKING |
25kgs
in drum
|
| HAZARD
CLASS |
|
| UN
NO. |
|
| OTHER
INFORMATION |
| Hazard Symbols: XN, Risk Phrases: 20/21/22, Safety
Phrases: 36 |
| GENERAL
DESCRIPTION OF BIPHENYL
|
| Biphenyl (also
called Diphenyl, but exactly one of two compounds. The other is o-phenylphenol,
used to prevent the growth of moulds ) is an aromatic hydrocarbon; molecule
structure is composed of two six-sided carbon rings connected at one carbon site
on each ring. Pure biphenyl is a toxic colourless crystalline solid with a
pleasant odour; melting point 70 C, boiling point 255 C, which gives plates or
monoclinic prismatic crystals; it is insoluble in water but soluble in ordinary
organic solvents. It is directly used in the preservation of citrus fruits as a
fungistat in transportation containers. It is a raw material for polychlorinated
diphenyls (PCB) in which chlorine replaces hydrogen in biphenyls. There are 209
chlorinated isomers of biphenyl theoretically. But PCBs are referred to the
biphenyl compounds with one to ten chlorine substitutions. PCBs are used
heat-transfer agents and as electric insulators that block the flow of electric
current across in electrical equipments. They are known as environmental
pollution materials which are accumulated in animal tissues and cause toxic
effects including carcinogenesis. Biphenyl is used as an intermediate for the
production of a wide range of organic compounds (e.g. emulsifiers, optical
brighteners, crop protection products, plastics), as a heat transfer medium
alone or with diphenyl ether in heating fluids, as a dyestuff carrier for
textiles and copying paper and as a solvent in pharmaceutical production. Aminodiphenyls
are used as rubber
antioxidants and intermediates for the synthesis of
organic compounds ( azo dyes and pharmaceuticals). Biphenyl derivatives
are used as an intermediate for the synthesis of organic
compounds including pharmaceuticals, antifungal agents,
optical
brightening agents and dyes.
Biphenyl compounds also used in luminescence
chemistry, spectrophotometric analysis, molecular
chemistry, and as a stating material
for organometallic-complexes. |
