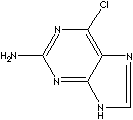
| 6-CHLOROGUANINE | ||
|
PRODUCT IDENTIFICATION |
||
| CAS NO. | 10310-21-1 |
|
| EINECS NO. | 233-686-7 | |
| FORMULA | C5H4ClN5 | |
| MOL WT. | 169.57 | |
|
H.S. CODE |
2933.99.9700 | |
| SMILES |
c12c(c(nc(n1)N)Cl)[nH]cn2 |
|
|
TOXICITY |
|
|
| SYNONYMS | 2-Amino-6-chloropurine; 6-Chloroguanine; 2-Amino-6-chlorpurine; | |
| 6-Chloro-1H-purin-2-amine; 6-Chloro-2-aminopurine; 6-Chloroguanine; 6-Chloro-7H-purin-2-ylamine; | ||
|
CLASSIFICATION |
Halogenopurine |
|
|
PHYSICAL AND CHEMICAL PROPERTIES |
||
| PHYSICAL STATE | white to off-white powder | |
| MELTING POINT | > 300 C | |
| BOILING POINT | ||
| SPECIFIC GRAVITY | ||
| SOLUBILITY IN WATER | Soluble | |
| pH | ||
| VAPOR DENSITY |
|
|
|
AUTOIGNITION |
|
|
|
NFPA RATINGS |
Health: 1; Flammability: 0; Instability: 0 | |
|
REFRACTIVE INDEX |
|
|
| FLASH POINT | ||
| STABILITY |
Stable under ordinary conditions |
|
|
GENERAL DESCRIPTION AND APPLICATIONS |
||
| Purine is a heterocyclic compound featured by a fused pyrimidine and imidazole
rings composed of carbon and nitrogen atoms. The simplest one is purine itself
and the two major purines are adenine(6-Aminopurine) and
guanine(2-Amino-6-hydroxypurine) which are two bases components of nucleic acid
and the nucleotides. Purine itself is not found in nature, but as substituted
purines such as methyled, hydroxyl and amino substituted. In addition to adenine
and guanine, a group of chemical compounds called purine base include
hypoxanthine (6-oxypurine), xanthine (2,6-dioxypurine), uric acid
(2,6,8-trioxypurine), and theobromine (3,7-dimethyl xanthine). Theophylline and
caffeine are a member of methylated purine family. Purines are biologically
important in In medicine and biological research.
http://journals.iucr.org/e/issues/2005/02/00/wk6041/ http://www.analytyka.com.mx/english/MSDS/A/A0566.htm http://kr.srd.yahoo.com |
||
| SALES SPECIFICATION | ||
|
APPEARANCE |
white to off-white powder | |
|
ASSAY |
99.0% min | |
| MELTING POINT | > 300 C | |
| TRANSPORTATION | ||
| PACKING |
|
|
| HAZARD CLASS | Not regulated | |
| UN NO. | ||
| OTHER INFORMATION | ||
| Hazard Symbols: XN , Risk Phrases: 22, Safety Phrases: 24/25 | ||