|
Phytosterol: various sterols naturally obtained from plants. It is chemically
similar to cholesterol which is obtained from animals. Sterol consists of three
fused cyclohexane rings with hydrophenanthrene ring arrangement; the side chains
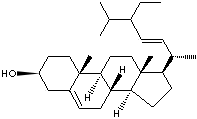
vary sterols (more than 40 phytosterols are known). Beta-sitosterol is the most
abundant phytosterol and others include campesterol, stigmasterol, ergosterol,
brassicasterol, delta-7-stigmasterol and delta-7-avenasterol. They are waxy,
clear solids soluble in most organic solvents but insoluble in water, and
contain one alcohol functional group. Sitosterols are considered to play roles
in lowering cholesterol in body (anticholesterolemic agent). stigmasterol is an
unsaturated plant sterol occurring in plant fats like such as calabar bean,
soybean oil, rape seed and cocoa butter. It is used as a starting material in
the manufacture of synthetic progesterone, a female sex hormone that plays an
important physiological role to regularize and rebuild changes to the body
caused by estrogen as well as in the luteal phase of the menstrual cycle. Its
levels vary during the menstrual cycle. In addition, progesterone is used as an
intermediate in the biosynthesis of androgens, estrogens, and the corticoids.
Synthetic compounds with progesterone are used in the prevention of miscarriage,
in the treatment of menstrual disorders. Ergosterol, also called provitamin D2,
is an unsaponifiable lipids; found in ergot, yeast, and other fungi; a white
crystalline compound insoluble in water and soluble in organic solvents. It is
converted to ergocalciferol (vitamin D2) upon irradiation by ultraviolet light
or electronic bombardment. vitamin D is either of two fat-soluble sterol-like
compounds with antirachitic activity, or both collectively: ergocalciferol
(vitamin D2) and activated cholecalciferol (vitamin D3). Ergocalciferol is
formed from the irradiation of ergosterol found in dietary supplements such as
fish liver oils, egg yolks, and fortified dairy products. However,
cholecalciferol is normally synthesized in the skin, where ultraviolet light
activates the compound 7-dehydrocholesterol. Vitamins D2 and D3 are about equal
in activity in all mammals. Deficiency of vitamin D can result in rickets in
children and osteomalacia in adults, while ingestion of excess levels can lead
to hypercalcemia, mobilization of calcium from bone, and renal dysfunction. |