|
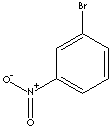
m-BROMONITROBENZENE
| ||
|
PRODUCT IDENTIFICATION |
||
| CAS NO. | 585-79-5 |
|
| EINECS NO. | 209-563-9 | |
| FORMULA | C6H4BrNO2 | |
| MOL WT. | 202.01 | |
|
H.S. CODE |
2904.90 | |
|
TOXICITY |
||
| SYNONYMS | 3-Bromo-1-nitrobenzene; | |
| 3-Bromonitrobenzene; 3-Nitrobromobenzene; 1-Bromo-3-nitrobenzene; | ||
| SMILES |
|
|
|
CLASSIFICATION |
||
|
PHYSICAL AND CHEMICAL PROPERTIES |
||
| PHYSICAL STATE | yellow crystalline powder | |
| MELTING POINT | 53 - 56 C | |
| BOILING POINT |
256 C | |
| SPECIFIC GRAVITY |
1.70 - 1.71 | |
| SOLUBILITY IN WATER | insoluble | |
| pH | ||
| VAPOR DENSITY |
| |
|
AUTOIGNITION |
| |
|
NFPA RATINGS |
Health: 2; Flammability: 1; Reactivity: 0 | |
|
REFRACTIVE INDEX |
| |
| FLASH POINT | 112 C | |
| STABILITY |
Stable under ordinary conditions | |
|
APPLICATIONS |
||
The prefix nitro- indicates the presence of NO2- radical, while nitrate refers
to any salt or ester of nitric acid or the NO3- anion. Nitroso- is the prefix
indicating presence of the group -NO and azo- is for -N=N- group. Some range of
organic compounds containing nitrogen include nitro compounds (RNO2 ), nitroso
compounds (RNO), amines (R3N ), amino acids, and natural alkaloids or
nucleotides. The nitrogen ion in nitro compounds is trigonally planar with 120°
angles. There are two resonance bonds so that the two oxygens are equivalent.
Nitro compounds are strongly basic due to electron withdrawing both inductively
and mesomerically. Historically, they
are abundant in dyes and explosives. Nitro compounds, organic hydrocarbons having one or more NO2
groups bonded via nitrogen to the carbon framework, are versatile intermediate
in organic synthesis.
m-Bromonitrobenzene is used as an intermediate for pharmaceuticals and other organic compounds |
||
| SALES SPECIFICATION | ||
|
APPEARANCE |
yellow crystalline powder | |
|
ASSAY |
99.0% min |
|
|
MELTING POINT |
53 - 56 C | |
| TRANSPORTATION | ||
| PACKING | 25kgs
in fiber drum | |
| HAZARD CLASS | ||
| UN NO. | ||
| OTHER INFORMATION | ||
| Hazard Symbols: T, Risk Phrases: 23/24/25/33, Safety Phrases: 28A/37/45 | ||
|
GENERAL DESCRIPTION OF NITROBENZENE |
||
|
Nitrobenzene (also called nitrobenzol ) is a colourless to pale yellow, oily, highly toxic liquid with the odour of bitter almonds. Nitrobenzene is manufactured commercially by nitration of benzene (also a common air pollutant) using a mixture of nitric and sulfuric acids. Commercially nitrobenzene can be either produced in a batch or by a continuous process. Both batch and continuous processes employing mixed nitric and sulfuric acids are used to make nitrobenzene. The continuous process is favored over the batch process because its production capacity is much larger, it has lower capital costs and it entails more efficient labor usage. Reactors for the continuous process also usually utilized lower nitric acid concentrations, are smaller with more rapid and efficient mixing, and therefore have higher reaction rates. Nitrobenzene undergoes nitration, halogenation, and sulfonation much more slowly than does benzene. It may be reduced to a variety of compounds, depending on the reaction conditions. Most nitrobenzene produced is reduced to aniline; smaller amounts are converted to azobenzene, hydrazobenzene (the intermediate for benzidine), and phenylhydroxylamine. Reduction of both the nitro group and the benzene ring affords cyclohexylamine. Nitrobenzene is used as a mild oxidizing agent in the syntheses of quinoline and fuchsin. Nitrobenzene is used to produce lubricating oils such as those used in motors and machinery. Nitrobenzene and its derivatives are used in the manufacture of dyes, drugs, pesticides, polisher, paint, and synthetic rubber. |
||