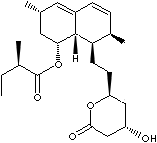
PRODUCT IDENTIFICATION
H.S. CODE
TOXICITY
Oral Mouse LD50: > 1,000 mg/kg
CLASSIFICATION
Anticholesteremic, Antihyperlipidemic, Antilipemic, Hydroxymethylglutaryl-CoA reductase inhibitor, Hypolipidemic, Lipid Regulator
EXTRA NOTES
PHYSICAL AND CHEMICAL PROPERTIES
AUTOIGNITION
NFPA RATINGS
Health: 1, Flammability: 0, Reactivity: 0
REFRACTIVE INDEX
EXTERNAL LINKS & GENERAL DESCRIPTION
Drug Information Portal (U.S. National Library of Medicine) - Lovastatin
PubChem Compound Summary - Lovastatin
Drug Bank - Lovastatin
KEGG (Kyoto Encyclopedia of Genes and Genomes) - Lovastatin
http://www.sigmaaldrich.com/
Lovastatin is a cholesterol lowering drug and competitive inhibitor of
HMG-CoA reductase, a rate limiting enzyme in cholesterol synthesis. Blocks the
production of mevalonate, a critical compound in the production of cholesterol
and isoprenoids. This product is a substrate of Pgp and CYP3A. It increases
cellular resistance to anticancer agents such as doxorubicin and induces
apoptosis in myeloma cells. The roles of Pgp and CYP3A, possible connection
between drug resistance, regulation of the mevalonate pathway, and
iosprenylation of signaling proteins in these observations remain to be
resolved. The product induces apoptosis in numerous cancer cell lines perhaps,
in part, by inhibiting the isoprenylation of Rho family GTPases. It causes cell
cycle arrest in G1 and G2/M phases.
American Chemical Society
Cholesterol-lowering drugs, like Merck's lovastatin, are a huge industry.
Pfizer's Lipitor is the world's top drug, with sales last year of more than $10
billion. And as a class, cholesterol- and triglyceride-lowering drugs were the
world's top category in 2004, with sales exceeding $30 billion, according to IMS
Health, a pharmaceutical market research firm.
Cholesterol is an essential blood component that is present in all body
cells. In particular, cholesterol serves to build the cell membranes of the
nerves and is found along the nervous system. The body acquires cholesterol
through diet or creates it in the liver. Critics of statin drugs claim that
because cholesterol is essential to life, it is perilous to artificially alter
its level in the blood.
Statins do induce a number of undesirable side effects in a minority of
patients. Former astronaut Duane E. Graveline wrote a book in which he blamed
Lipitor for episodes of memory loss. Statins may also set off muscle disorders
such as myositis or rhabdomyolysis. Myositis may cause no more than muscle pain,
but rhabdomyolysis is a form of muscle breakdown that can be fatal. Bayer agreed
to remove its statin Baycol from the market in 2001 after it was linked to more
than 50 cases of rhabdomyolysis. (source:
)
Lovastatin: A lactone metabolite isolated from the fungus Aspergillus terreus with cholesterol-lowering and potential antineoplastic activities. Lovastatin is hydrolyzed to the active beta-hydroxyacid form, which competitively inhibits 3-hydroxyl-3-methylgutarylcoenzyme A (HMG-CoA) reductase, an enzyme involved in cholesterol biosynthesis. In addition, this agent may induce tumor cell apoptosis and inhibit tumor cell invasiveness, possibly by inhibiting protein farnesylation and protein geranylgeranylation, and may arrest cells in the G1 phase of the cell cycle. The latter effect sensitizes tumor cells to the cytotoxic effects of ionizing radiation (source: )
Local
statins (or hydroxymethylglutaryl coenzyme A reductase inhibitors)
|
Statin |
CAS RN. |
|
Mevastatin |
73573-88-3 |
| Lovastatin | 75330-75-5 |
| Simvastatin | 79902-63-9 |
| Pravastatin | 81093-37-0 |
| Fluvastatin | 93957-54-1 |
| Fluvastatin Sodium | 93957-55-2 |
| Atorvastatin | 134523-00-5 |
| Atorvastatin Calcium | 134523-03-8 |
| Cerivastatin Sodium | 143201-11-0 |
| Cerivastatin | 145599-86-6 |
| Pitavastatin | 147511-69-1 |
| Rosuvastatin | 287714-41-4 |
APPEARANCE
ASSAY
MELTING POINT
205 - 207 C
LOSS ON DRYING
0.3% max
IMPURITY
Total
impurity: 1.0%
max
Individual Impurity: 0.3% max
0.2% max
20ppm max
Hazard OVERVIEW
GHS
PICTOGRAMS

HAZARD STATEMENTS
H302 Harmful if swallowed
SAFETY PHRASES
22 -24/25