PRODUCT IDENTIFICATION
H.S. CODE
TOXICITY
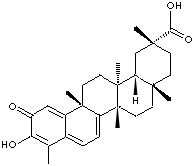
SMILES
C12=CC(=O)C(=C(C2=CC=C2[C@]1(CC[C@@]1([C@@]2(CC[C@ @]2([C @H] 1C[C@](C(=O)O)(CC2)C)C)C)C)C)C)O
CLASSIFICATION
Triterpenoid, Anti-Inflammatory
EXTRA NOTES
Isolated from Tripterygium wilfordii; potent inhibitory activity on both Kir2.1 and ERG1 potassium channels, leading to long qt syndrome.
PHYSICAL AND CHEMICAL PROPERTIES
red crystalline powder
190~ 195 C
Insoluble
Soluble in most nonpolar solvents, DMSO, ethanol.
AUTOIGNITION
NFPA RATINGS
Health Hazard: 2 Fire: 0 Reactivity Hazard:0
REFRACTIVE INDEX
GENERAL DESCRIPTION & EXTERNAL LINKS
Wikipedia Linking - Quinone methide
Google Scholar Search - Celastrol
Drug Information Portal (U.S. National Library of Medicine) - Tripterine
PubChem Compound Summary - Celastrol
http://www.ebi.ac.uk/ - Celastrol
http://www.ncbi.nlm.nih.gov/ - Celastrol
http://cancerres.aacrjournals.org/
Celastrol has been effectively used in the treatment of autoimmune diseases, asthma, chronic inflammation, and neurodegenerative disease. Under in vitro conditions, Celastrol was found to inhibit cancer cell proliferation and induce leukemic cell death; however, the molecular mechanism involved still remains unclear.
Chinese Medical Journal
Celastrol is a major active component of Tripterygium wilfordii named “Thunder God Vine”, which is widely used to treat rheumatoid arthritis in China. The present study aims to demonstrate that celastrol has potent anticancer activity against glioma in vitro and in vivo.
Local
Tripterine (celastrol) is a pentacyclic triterpenoid compound isolated from the Chinese Tripterygium wilfordii Hook. It has an activity of immunosuppressive and anti-inflammatory. Triterpenes antioxidants are not common, but tripterine's antioxidant potency is equivalent to about 15 times that of alpha-tocopherol.
APPEARANCE
red crystalline powder
190~ 195 C
HAZARD OVERVIEW
Toxic if swallowed. May be harmful if inhaled. May cause respiratory tract irritation. May be harmful if absorbed through skin. May cause skin irritation. May cause eye irritation.
GHS
Warning
PICTOGRAMS

HAZARD STATEMENTS
H301
P STATEMENTS
P301 + P310
![]() T
T
RISK PHRASES
25
SAFETY PHRASES
45