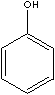
PHENOL
PRODUCT IDENTIFICATION
108-95-2
94.11
H.S. CODE
CLASSIFICATION
PHYSICAL AND CHEMICAL PROPERTIES
Clear to light pink crystals
MELTING POINT
40 - 43 C
8 g/100 ml (easily soluble in alcohol)
AUTOIGNITION
NFPA RATINGS
REFRACTIVE INDEX
GENERAL DESCRIPTION & APPLICATIONS
Pure phenol is a white crystalline solid that melts at 41 C, boils at 182 C. It is moderately soluble in water and is soluble in ethanol and ether. Phenols form stronger hydrogen bonds than aliphatic alcohols and. Phenols are more soluble in water than alcohols and have higher boiling points. Many phenols have a sharp, spicy odour, but phenol smells bland and sweetish. It is highly toxic and caustic. Some phenols interfere with the endocrine system and disrupt the function of hormones. They have antiseptic property and are used in formulating disinfectants, deodorizers, and pesticides.
Phenol is synthesized by either the hydrolysis of chlorobenzene ( Raschig process) or the oxidation of cumene in air to form cumene hydroperoxide, which is then cleaved into phenol and acetone, a valuable by-product ( Cumene process ). Phenols are acidic and react with strong bases to form alkali-metal salts known as phenoxides. The most important reaction of phenol is its condensation (which produces water as a side effect) with formaldehyde. Commercially main derivatives from phenol are ; Bisphenol A ( > Polycarbonate and Epoxy Resins), Phenolic Resins ( > Moulding Binders, Insulation Wool), Cyclohexane ( > Caprolactam), Alkylphenols ( > Surfactants), Salicyclic Acid ( > Pharmaceuticals). Phenol is an important parent material in the production of thermosetting resins, drugs, dyes, pesticides, and explosives.
BPA GRADE
MOLTEN COLOR
10 max (Pt/Co Scale)
WATER
0.03% max
Fe
1ppm max
SOLUTION CLARITY
Clear (in water)
0.05% max
RESIN GRADE
PURITY
99.7% min
MOLTEN COLOR
20 max (Pt/Co Scale)
WATER
0.1% max
SOLUTION CLARITY
Clear (in water)
TRANSPORTATION
1671