| CAS
NO. |
446-35-5 |
|
| EINECS
NO. |
207-167-0 |
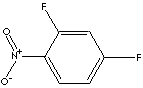
| FORMULA |
C6H3F2NO2 |
| MOL
WT. |
159.09 |
| H.S.
CODE |
|
| TOXICITY |
Oral rat LD50: 200 mg/kg |
| SYNONYMS |
2,4-Difluoro-1-nitrobenzene;
|
|
1-Nitro-2,4-difluorobenzene; 1,3-Difluoro-4-nitrobenzene;
|
|
SMILES |
|
|
CLASSIFICATION
|
|
|
PHYSICAL AND CHEMICAL PROPERTIES
|
| PHYSICAL
STATE |
clear
to yellowish liquid
|
| MELTING POINT |
9
- 10 C |
| BOILING
POINT |
203 - 204 C |
| SPECIFIC GRAVITY |
1.455 |
| SOLUBILITY
IN WATER |
Insoluble |
| pH |
|
| VAPOR DENSITY |
5.49 |
| AUTOIGNITION
|
|
| NFPA RATINGS |
Health: 2; Flammability: 2; Reactivity: 0 |
| REFRACTIVE
INDEX
|
|
| FLASH
POINT |
90
C
|
| STABILITY |
Stable under ordinary conditions |
|
APPLICATIONS
|
The prefix nitro- indicates the presence of NO2- radical, while nitrate refers
to any salt or ester of nitric acid or the NO3- anion. Nitroso- is the prefix
indicating presence of the group -NO and azo- is for -N=N- group. Some range of
organic compounds containing nitrogen include nitro compounds (RNO2 ), nitroso
compounds (RNO), amines (R3N ), amino acids, and natural alkaloids or
nucleotides. The nitrogen ion in nitro compounds is trigonally planar with 120°
angles. There are two resonance bonds so that the two oxygens are equivalent.
Nitro compounds are strongly basic due to electron withdrawing both inductively
and mesomerically. Historically, they
are abundant in dyes and explosives. Nitro compounds, organic hydrocarbons having one or more NO2
groups bonded via nitrogen to the carbon framework, are versatile intermediate
in organic synthesis.
- Michael
addition
- Reduction
- Henry
Reaction (Nitro-aldol reaction)
- Nef
reaction
- O-Alkylation
- Cycloaddition
- Substitution, Elimination, Conversion
reaction
- Alkylation,
Acylation, and Halogenation
|
| SALES
SPECIFICATION |
|
APPEARANCE
|
clear
to yellowish liquid
|
| PUIRTY |
99.0%
min
|
| TRANSPORTATION |
| PACKING |
|
| HAZARD CLASS |
6.1
(Packing Group: II) |
| UN
NO. |
2810 |
| REMARKS |
|
Hazard
Symbols: T, Risk Phrases: 23/24/25-33, Safety Phrases:
36/37/39-45 |
