PRODUCT IDENTIFICATION
CLASSIFICATION
PHYSICAL AND CHEMICAL PROPERTIES
REFRACTIVE INDEX
APPLICATIONS
Ether is any of a number of organic compounds characterized by an oxygen atom joined with single bonds by two carbon atoms that are part of hydrocarbon groups. The general formula is R-O-R', where R and R' are alkyl or aromatic groups. Ethers are formed by the condensation of two alcohols by heating with sulfuric acid; the reaction is one of dehydration. Ethers can be prepared from alkyl halide reacted with metallic alkoxide (called Williamson synthesis). Ethers are similar to alcohols but are generally less dense, less soluble in water, and have lower boiling points. They are relatively unreactive, which makes them valuable solvents. But ethers will be cleaved at high temperatures by concentrated hydrogen halides. Ethers have relatively low boiling point compare to alkanes as they don't form hydrogen bonds each other. Ethers are more lipophilic than esters [R-C(=O)-O-R']or amides [RCO-NH2]. Ethers are widely used as solvents for various organic reactions because they are relatively the least reactive among common organic compounds except alkanes and fluorocarbons. The common reaction of ethers is cleavage of the C–O bond by strong acids either in linear chain or cyclic structure. Ethers in which oxygen is bonded to primary and secondary alkyl groups can form peroxide compounds in the presence of gaseous oxygen due to two unpaired electrons in oxygen. Ethers can act as Lewis bases in chemical reactions. Commonly, ethers are named simply in listing the alkyl groups in alphabetical order or alkane order such as ethyl methyl ether or methyl ethyl ether, which is methoxyethane in IUPAC nomenclature ( the formula of "alkoxyalkane" ). When ether is a parts of complex molecule or aromatic derivatives, it is described as an alkoxy substituent such as methoxybenzene ( trivial name is anisole). The methoxy prefix indicates the function methyl group joined by single bonds to an oxygen atom, with the general formula -O-CH3. Cyclic ethers have ring structure where the oxygen has become part of the ring. The term of epoxide indicate three membered cyclic ether (also called oxirane) in which an oxygen atom is joined to each of two carbon atoms that are already bonded to each other; four membered cyclic ether is called oxetane; five membered cyclic ether, furan (or oxolane); six membered cyclic ether, pyran (also called oxane) respectively. Their unhindered oxygen atom carries two unshared pairs of electrons - a structure which favors the formation of coordination complexes and the solvation of cations. Cyclic ethers are used as important solvents, as chemical intermediate and as monomer for ring-opening polymerization. Crown Ether is a macrocyclic polyether whose structure contains hydrogen, carbon and oxygen atoms. Each oxygen atoms are confined between two carbon atoms and exhibits a conformation with a hole (accordingly called "crown"). Anisole is one of the simplest aromatic compound to which ether group is linked. But it is different with aromatic compounds like furan where the oxygen is a part of the ring. Anisole, C6H5OCH3 (methyl phenyl ether), is a clear liquid that is soluble in ether and alcohol; insoluble in water; boiling point 155 C. Anisole and its derivatives are used as solvents and in perfumery. Anisole can be obtained from anise seed. Anisic acid, p-methoxybenzoic acid, is a part of cresol class antiseptic compounds. It is also used as an insect repellent and ovicide. Anisole, anisic acid, and their derivatives are also widely used in chemical reaction as intermediates to obtain target materials such as dyes, pharmaceuticals, perfumes, photoinitiators and agrochemicals.
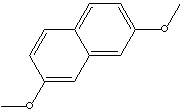
Naphthalenediols and their derivatives are used directly in making several dyes and are converted into numerous dye intermediates, as well as into tanning agents, antioxidants, and antiseptics.
APPEARANCE
98.0% min
Hazard Symbols: , Risk Phrases: , Safety Phrases: 24/25