|
n-BUTYL BROMIDE | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
PRODUCT IDENTIFICATION |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS NO |
109-65-9 |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| EINECS NO. |
203-691-9 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
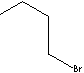
| FORMULA | CH3(CH2)3Br | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MOL WT. |
137.02 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| H.S. CODE | 2903.30 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TOXICITY | Oral rat LD50: 2761 mg/kg | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SYNONYMS | 1-Bromobutane; 1-Butyl bromide; | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
SMILES |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
CLASSIFICATION |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
PHYSICAL AND CHEMICAL PROPERTIES |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PHYSICAL STATE |
clear to yellow liquid | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MELTING POINT | -112 C | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BOILING POINT | 101 - 102 C | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SPECIFIC GRAVITY | 1.28 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SOLUBILITY IN WATER | Very slightly soluble | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| AUTOIGNITION | 265 C | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| pH | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| VAPOR DENSITY | 4.72 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| NFPA RATINGS | Health: 2 Flammability: 3 Reactivity: 0 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
REFRACTIVE INDEX |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FLASH POINT |
20 C | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| STABILITY | Stable under ordinary conditions. Oxidizes in light | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
APPLICATIONS |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Halogenoalkanes, also known as haloalkanes or alkyl halides, are organic
compounds in which one or more hydrogen atoms in an alkane have been replaced by
halogen atoms, fluorine, chlorine, bromine or iodine. In carbon-halogen bond,
halogens have significantly greater electronegativities than carbon except
iodine. In result, this functional group is polarized so that the carbon is
electrophilic and the halogen is nucleophilic. Halogenoalkanes are can be
classified depending on the halogen atom position on the chain of carbon atoms.
The carbon which is attached with the halogen atom is linked up with only one
other alkyl group in primary halogenoalkanes, whereas directly linked up with
two and three other alkyl groups in secondary halogenoalkanes and tertiary
halogenoalkanes respectively. In some case, primary halogenoalkanes are counted
even though there are no alkyl groups attached to the carbon with the halogen on
it. Three characteristics provide important influences on the chemical behavior
of halogenoalkanes, these are electronegativity, covalent bond strength and the
relative stability of the corresponding halide anions. Fluoroalkanes have the
strongest of the carbon-halogen covalent bonds so that they are unreactive. This
is stronger single bond than a carbon-carbon bond. The carbon-chlorine covalent
bond is slightly weaker than a carbon-carbon bond, and the bonds to the other
halogens are weaker. The stability may be estimated from the relative acidities
of the H-X acids. All the hydrohalic acids are very strong, but with small
differences in the direction HCl < HBr < HI, with the exception of HF.
Halogenoarenes, also called haloarene, or aryl Halide, are an organic compound
in which one or more hydrogen atoms in an aromatic ring have been replaced by
halogen atoms. The Haloarenes exhibit many differences compare to haloalkanes in
the method of preparation and their chemical and phisical properties.
Haloalkanes are used in as refrigerants, solvents, blowing agents, aerosol
propellants, fire extinguishing media , and in semiconductor device fabrication.
One of big consumption of halogenoalkanes (properly speaking, halogenoalkenes)
is as a raw material to prepare plastics such as PVC [poly(chloroethene)] from
chloroethene and PTFE [poly(tetrafluoroethene)] from tetrafluoroethene.
Halogenoalkanes and halogenoarenes react with lots of compounds resulting in a
wide range of different target substances. They are useful intermediates in
making other organic compounds.
1-Bromobutane, also known as n-butyl bromide is a primary alkyl halide, with the formula CH3CH2CH2CH2Br . It is colorless liquid, insoluble in water, but soluble in ethanol and ether; melting point -112 C; boiling point 101 - 102 C. It is used as an alkylating agent to introduce the butyl groups to form carbon-carbon bonds in organic synthesis. They are also used as intermediate to form alkylated amines and alkylated metallic compounds. The end products include pharmaceuticals, insecticides, quaternary ammonium compounds, flavours and fragrances. Some examples of C4 bromide compounds include
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SALES SPECIFICATION | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
APPEARANCE |
clear to yellow liquid | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ASSAY (G.C) |
99.0% min | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
COLOR, APHA |
50 max | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
1-BUTANOL |
0.5% max | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
WATER |
0.1% max | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TRANSPORTATION | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PACKING | 250kgs in drum | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HAZARD CLASS | 3 (Packing group: III) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| UN NO. |
1126 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
REMARKS |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||