PRODUCT IDENTIFICATION
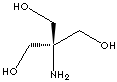
CLASSIFICATION
Buffer, Alkalize, Antibacterial, Pharmaceutical Vehicle, Excipient, Good's buffer, Amino alcohol
EXTRA NOTES
An organic amine proton acceptor.
EPA
Pesticide Chemical Code 083901
PHYSICAL AND CHEMICAL PROPERTIES
REFRACTIVE INDEX
EXTERNAL LINKS & GENERAL DESCRIPTION
Drug Information Portal (U.S. National Library of Medicine) - Tris
PubChem Compound Summary - Tromethamine
http://www.ebi.ac.uk/chebi/ - Tromethamine
http://www.ncbi.nlm.nih.gov/ - Tromethamine
http://www.sigmaaldrich.com/
The pH values of all buffers are temperature and concentration dependent. For
Tris buffers, pH increases about 0.03 unit per °C decrease in temperature, and
decreases 0.03-0.05 unit per ten-fold dilution. For precise applications, use
a carefully calibrated pH meter with a glass/calomel combination electrode.
http://msdssearch.dow.com/
TRIS AMINO is well-known worldwide as a buffer in biochemical reactions. It can be used to buffer anywhere within the pH range of 7 to 9 by utilizing a combination of the free base and its salts; the hydrochloride salt is especially popular for this purpose. In fact, a buffer solution containing TRIS AMINO and its hydrochloride salt at a molar ratio of 1:3 is considered a standard buffer solution for use in the physiological pH range of 7.3 to 7.5
Local: Buffer is a substance, generally a solution, that can keep its pH constant, despite the addition of strong acids or strong bases and external influences of temperature, pressure, volume, redox potential. Buffer prevents change in the concentration of another chemical substance, e.g., proton donor and acceptor systems that prevent marked changes in hydrogen ion concentration (pH). Many acid-base reactions take place in living organisms. However, for organisms to perform certain vital functions, the body fluids associated with these functions must maintain a constant pH. For example, blood must maintain a pH of close to 7.4 in order to carry oxygen from the lungs to cells; blood is therefore a powerful buffer. The commonest buffer in chemical solution systems is the acid-base buffer.
- Bicarbonate buffer; a buffer system composed of bicarbonate ions and dissolved carbon dioxide; in the body, this system is an important factor in determining the pH of the blood as the concentration of bicarbonate ions is regulated by the kidneys and of carbon dioxide by the respiratory system.
- Cacodylate buffer; one containing an organic arsenical salt, used in preparing fixatives for electron microscopy.
- Phosphate buffer, a buffer system composed of KH2PO4 and Na2HPO4; in the body, it is important in regulating the pH of the renal tubular fluids; when 0.025 molal (equimolal of the potassium and sodium salts), the pH is 6.865 at 25 C.
- Protein buffer, a buffer system involving proton donor and proton acceptor groups of the amino acid residues of proteins.
- TRIS buffer (tromethamine): an amine base used intravenously as an alkalizer for the correction of metabolic acidosis. The pH values of all buffers are temperature- and concentration-dependent. For Tris buffers, pH increases about 0.03 unit per C temperature decrease, and decreases 0.03-0.05 unit per ten-fold dilution.
- Veronal buffer; a barbital buffer commonly used in the preparation of fixatives for electron microscopy.
Tromethamine is used as an intermediate for the preparation of surface active agents, vulcanization accelerators, and pharmaceuticals, and used as a titrimetric standard.
|
|
APPEARANCE
99.9% min
168 - 172 C
WATER INSOLUBLES
0.1% max
Cl
5ppm max
LOSS ON DRYING
0.5% max
HEAVY METALS
5ppm max
CLARITY
clear (in 40% water)
pH
10 - 11.5
SULFATE
5ppm max
HAZARD OVERVIEW
GHS
PICTOGRAMS

HAZARD STATEMENTS
H315:
Causes skin irritation
H335: May cause respiratory
irritation
H319: Causes serious eye irritation
PRECAUTIONARY STATEMENTS
P261:
Avoid breathing dust/fume/gas/mist/vapors/spray
P280: Wear
protective gloves/protective clothing/eye protection/face protection
P302
+ P352: IF ON SKIN: Wash with plenty of soap and water
P305
+ P351 + P338: IF IN EYES: Rinse cautiously with water for
several minutes. Remove contact lenses, if present and easy to do.
Continue rinsing
![]() Xi
Irritant
Xi
Irritant
RISK PHRASES
36/37/38: Irritating to eyes, respiratory system and skin.
SAFETY PHRASES
26:
In case of contact with eyes, rinse immediately with plenty
of water and seek medical advice.
37/39: Wear suitable
gloves and eye/face protection.
PRICE
INFORMATION