|
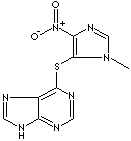
AZATHIOPRINE |
| 6-((1-Methyl-4-nitroimidazol-5-yl)thio)purine; 6-((1-Methyl-4-nitro-1H-imidazol-5-yl)thio)purine; Imuran; Imurek; Imurel; Methylnitroimidazolylmercaptopurine; Muran; 6-(1'-Methyl-4'-nitro-5'-imidazolyl) mercaptopurine; 6-(1-Methyl-4-nitroimidazol-5-ylthio)purin; 6-(1-Methyl-p-nitro-5-imidazolyl)thiopurine; 6-1'-Methyl,4'-nitro 5'-imidazolyl mercaptopurine; Azamun; Azamun; Azanin; Azasan; Azathioprin; Azathioprinum; Azathioprinum; Azatioprin; Azatioprina; Azothioprine; 6-(3-Methyl-5- nitroimidazol -4-yl)sulfanyl-7H-purine; |
|
|
| PRODUCT IDENTIFICATION |
|
|
CAS RN |
446-86-6, 55774-33-9 (hydrochloride) |
|
EINECS RN |
207-175-4 |
|
FORMULA |
C9H7N7O2S |
|
MOLE WEIGHT |
277.26 |
|
CLASSIFICATION |
Immunosuppressant / Antirheumatic Agent / Mercaptopurine |
|
|
| PHYSICAL AND CHEMICAL PROPERTIES |
|
|
PHYSICAL STATE |
pale yellow crystalline power |
|
MELTING POINT |
243 C (Decomposes) |
|
BOILING POINT |
|
|
DENSITY |
|
|
SOLUBILITY IN WATER |
Insoluble (very slightly soluble in ethanol, soluble in dilute alkali solution, sparingly soluble in dilute mineral acids) |
|
pH |
|
|
VAPOR DENSITY |
|
|
REFRACTIVE INDEX |
|
|
FLASH POINT |
|
|
|
| STABILITY AND REACTIVITY | |
| STABILITY | Stable under normal conditions |
|
INCOMPATIBLE MATERIALS |
Strong oxidizing agents. |
| DECOMPOSITION PRODUCTS |
Carbon oxides. nitrogen oxides, Sulphur oxides. |
| POLYMERIZATION | Has not been reported |
|
NFPA RATINGS |
Health: 2 Flammability: 1 Reactivity: 0 |
|
|
| SAFETY |
|
|
HAZARD NOTES |
Toxic. May cause cancer. Harmful if swallowed. Irritating to eyes, respiratory system and skin. |
|
EYE |
Irritating to eyes. |
|
SKIN |
Irritating to skin. |
|
INGESTION |
Harmful if swallowed. |
|
INHALATION |
Irritating to eyes, respiratory system. |
| MAIN RISKS AND TARGET ORGANS | Azathioprine is a myelotoxic and hepatotoxic immunosuppressive agent. Bone marrow and liver are the main targets but gastrointestinal tract, kidney, lungs, CNS and skin may also be affected. Transient gastroenteritis may be observed with massive overdose. Leukopenia is the main toxic effect which may occur during azathioprine therapy and in the overdose patients. Liver and kidney function tests may be altered but usually returned to normal after discontinuation of the drug. (http://www.inchem.org/) |
|
|
| TRANSPORT & REGULATORY INFORMATION |
|
|
UN NO. |
|
| HAZARD CLASS |
|
| PACKING GROUP |
|
| HAZARD SYMBOL |
T |
|
RISK PHRASES |
45-22-36/37/38 |
|
SAFETY PHRASES |
53-22-26-36/37-45 |
|
|
| EXTERNAL LINKS & GENERAL INFORMATION | |
|
Azathioprine is used in the treatment of several different types of rheumatic disease, including rheumatoid arthritis and systemic lupus erythematosus (SLE). Azathioprine has effects on the immune system (the body's own defence system). One of its actions is to reduce the activity of the immune system, so it is always used with care. Doctors may prescribe it for people on steroid treatment so their steroid dose can be smaller. (http://www.arc.org.uk/) Azathioprine is used as an adjunct for the prevention of the rejection of kidney allografts. The drug is used in conjunction with other immunosuppressive therapy including local radiation therapy, corticosteroids, and other cytotoxic agents. Azathioprine may be used for the treatment of conditions which involve derangement of the immune system including chronic active hepatitis, severe rheumatoid arthritis, systemic lupus erythematosus, dermatomyositis, pemphigus vulgaris, polyarteritis nodosa, acquired haemolytic anaemia, Crohn's disease and idiopathic thrombocytopenia. (Dollery, 1991; McEvoy, 1993). (http://www.inchem.org/)The main uses of azathioprine include:
Azathioprine is metabolised to mercaptopurine and then to thioinosinate which interferes with purine metabolism. Mercaptopurine interacts with allopurinol. The dose of azathioprine should be reduced if allopurinol is prescribed simultaneously. The most common side-effects of azathioprine are myelosuppression and hepatotoxicity. The summary of product characteristics should be consulted before prescribing this drug. (http://www.gpnotebook.co.uk/) Pharmacological actions:
|
|
|
| SALES SPECIFICATION |
|
|
APPEARANCE |
pale yellow crystalline power |
| PURITY |
98.0% min |
|
|
| PRICE INFORMATION |