|
DIPHENYL GUANIDINE | ||
|
PRODUCT IDENTIFICATION |
||
| CAS NO. | 102-06-7 24245-27-0 (monohydrochloride) 52392-53-7 (hydrochloride) |
|
| EINECS NO. | 203-002-1 | |
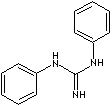
| FORMULA | C6H5NHC(=NH)NHC6H5 | |
| MOL WT. | 211.27 | |
|
H.S. CODE |
2925.29.1800 | |
|
TOXICITY |
Oral rat LD50: 375 mg/kg | |
| SYNONYMS | 1,3-Diphenylguanidine; DPG; 1,3-Difenylguanid; | |
| Melaniline; N,N'-Diphenylguanidine; Nocceler D; s-Diphenylguanidine; Dwufenyloguanidyna; Melaniline; Vulkacit D; (Other CAS RN: 25323-69-7; 33505-70-3; 39291-21-9; 55556-10-0); | ||
| SMILES | c1(NC(Nc2ccccc2)=N)ccccc1 | |
|
CLASSIFICATION |
Vulcanization accelerator |
|
|
PHYSICAL AND CHEMICAL PROPERTIES |
||
| PHYSICAL STATE | White to pale pink powder | |
|
MELTING POINT |
148 C | |
| BOILING POINT | 170 C (Decomposes) | |
| SPECIFIC GRAVITY | 1.15 | |
|
SOLUBILITY IN WATER |
1 mg/ml | |
| VAPOR DENSITY | ||
| pKa | 10.12 (Dissociation Constant at 20 C) | |
| log Pow | 2.89 (Octanol-water) | |
| VAPOR PRESSURE | 8.45E-06 (mmHg at 25 C) | |
| HENRY'S LAW | 7.12E-12 (atm-m3/mole at 25 C) | |
| OH RATE | 8.53E-11 (cm3/molecule-sec at 25 C Atmospheric ) | |
| AUTOIGNITION |
| |
|
REFRACTIVE INDEX |
| |
| NFPA RATINGS | Health: 2; Flammability: 0; Reactivity: 0 | |
| FLASH POINT |
| |
| STABILITY | Stable, Sensitive to moisture | |
|
GENERAL DESCRIPTION & APPLICATIONS |
||
|
Guanidine,
also called carbamidine, is a strongly alkaline and
water-soluble
compound, NHC(NH2)2 It is formed by
the oxidation of guanine in urine as a normal product
of protein metabolism in the body. In industry, guanidine,
containing nitrogens and N=C solid bond, and its modified
derivatives are versatile intermediates used
in the manufacture of plastics, resins, rubber chemicals,
nitroguanidines (explosives), photo
chemicals, fungicides, and disinfectant. It has also
biotechnological application of protein separation,
purification and as a protein denaturant. It can be
used as an oxygen scavenger to prevent corrosion
damage. It is used as a component of rocket propellants
because it produce a large amount of heat
when burned.
Diphenylguanidine is one of common accelerators for the vulcanization of rubber in combination with thiazoles and sulfenamides. Though it does not show better activity than thiuram and dithiocarbamates, it has better stability. It is used as a complexing agent for the detection of metals and organic bases. It is used as a secondary foam stabilizer in the silicoflouride foam process. http://www.faqs.org/ http://www.epa.gov/ http://www.astm.org/ |
||
| SALES SPECIFICATION | ||
|
APPEARANCE |
White to pale pink powder | |
| ACTIVE CONTENT |
96.0% min | |
| MELTING POINT |
146 C min | |
|
ASH |
0.3% max | |
| HEAT LOSS |
0.3% max | |
| FINENESS |
99.9% (100 mesh) | |
| COPPER (as Cu) |
0.001% max | |
| MANGANESE (as Mn) |
0.0001% max | |
| NITROGEN |
19.0% (Typical) | |
| PARAFIN OIL |
1.0 -2.0% | |
| TRANSPORTATION | ||
| PACKING | III | |
| HAZARD CLASS | 9 | |
| UN NO. | 8027 | |
|
SAFETY INFORMATION |
||
Hazard Symbols: XN N, Risk Phrases: 22-36/37/38-51/53-62, Safety Phrases: 26-36/37/39-61 |
||
| GENERAL DESCRIPTION OF ACCELERATOR | ||
|
Sulfur combines with nearly all elements. Sulfur forms ring and chain structures as it is the second only to carbon in exhibiting catenation. The 8-membered ring and shorter chain structure of sulfur molecule is important in vulcanization process which individual polymers are linked to other polymer molecules by atomic bridges. This process produces thermoset materials which are cross-linked and irreversible substances. The term thermoplastic is for high molecular weight polymers which can undergo melting-freezing cycle. Thermosets are not melted and re-molded on heating after cured. The split of sulfur 8-membered ring structure into shorter chains provides rubber vulcanization process. The split are liked with cure sites (some of the solid bonds in the molecule) on rubber molecules, resulting in forming sulfur bridges typically between 2 and 10 atoms long. Vulcanization makes rubber harder, more durable and more resistant to heating, aging and chemical attacks. The number of sulfur atoms in the sulfur bridges varies physical properties of the end products. Short bridges containing one or two sulfur atoms offer heat resistance and long bridges offer flexible property. Vulcanization can also be accomplished with certain peroxides, gamma radiation, and several other organic compounds. The principal classes of peroxide cross-linking agents are dialkyl and diaralkyl peroxides, peroxyketals and peroxyesters. Other vulcanizing agents include amine compounds for the cross-linking of fluorocarbon rubbers, metal oxides for chlorine-containing rubbers (notably zinc oxide for chloroprene rubber) and phenol-formaldehyde resins for the production of heat-resistant butyl rubber vulcanizates. Accelerator, in the rubber industry, is added with a curing agent to speed the vulcanization. Accelerators contain sulfur and nitrogen like derivatives of benzothiazole and thiocarbanilides. The popular accelerators are sulfenamides (as a delayed-action accelerators), thiazoles, thiuram sulfides, dithocarbamates and guanidines. There are some types of rubber accelerators. They are used in combination with each other in accordance with vulcanizing and/or acid-base conditions. Some examples classified by chemical structure are as below;
|
||
|
PRICE INFORMATION |
||