| CAS NO. |
123-30-8 |
|
| EINECS NO. |
204-616-2 |
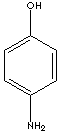
| FORMULA |
H2NC6H4OH |
| MOL WT. |
109.13 |
|
H.S. CODE
|
2922.29 |
|
TOXICITY
|
Oral rat LD50: 375 mg/kg |
| SYNONYMS |
4-Aminophenol p-Hydroxyaniline Paranol C.I.76550 |
|
4-Amino-1-hydroxybenzene Azol Certinal 4-Hydroxyaniline 4-Aminobenzenol |
| DERVATION |
p-Nitrophenol or Nitrobenzene |
|
CLASSIFICATION
|
|
|
PHYSICAL AND CHEMICAL PROPERTIES
|
| PHYSICAL STATE |
White or reddish crystal turns to dark on exposure to light
|
| MELTING POINT |
190 C |
| BOILING POINT |
284 C (Decomposes) |
| SPECIFIC GRAVITY |
|
| SOLUBILITY IN WATER |
1.5 g/100 ml |
| pH |
|
| VAPOR DENSITY |
|
| AUTOIGNITION |
|
|
NFPA RATINGS
|
Health: 2 Flammability: 0 Reactivity: 0 |
|
REFRACTIVE INDEX
|
|
| FLASH POINT |
|
| STABILITY |
Stable under normal condition. But discolor on exposure to light |
|
APPLICATIONS
|
|
The intermediate for paracetamol
(analgesic), 4-aminosalicylic acid (
tuberculostatic), sulphur dyes, rubber antioxidant, photo developer.
|
| SALES SPECIFICATION |
|
APPEARANCE
|
White to reddish crystal turns to darken on exposure to light
|
| ASSAY |
98.0% min
|
| Fe |
0.03% max
|
| MOISTURE |
1.0% max
|
| TRANSPORTATION |
| PACKING |
40kgs in Bag, 17mts in Container |
| HAZARD CLASS |
6.1 |
| UN NO. |
2512 |
| PRICE INDICATION |
| USD3.50/kg CFR by sea for the quantity 1,000kgs shipment. |
| OTHER INFORMATION |
|
SHELF LIFE : 3months
Hazard Symbols: XN N, Risk Phrases: 20/22/40/50/53, Safety Phrases: 28/36/37/60/61
|
|
GENERAL DESCRIPTION OF AMINE
|
Amine is a group of basic organic compounds derived by replacement of one (primary amines), two (secondary amines), or three (tertiary amines) of the hydrogen atoms in ammonia (NH3) by alkyl or aryl groups. Tertiary amines combine with one molecular proportion of an alkyl iodide to form quaternary ammonium salts in which a central nitrogen atom is joined to four organic radicals and one acid radical used as a corrosion inhibitor, emulsifying and antiseptic agents. Amines, like ammonia, are weak bases because the unshared electron pair of the nitrogen atom can form a coordinate bond with a proton. Amines react with acids to give salts and with acid anhydrides (or or ester ) to form amides. They react with halogenoalkanes to form longer chains. Low molecular amine names are formed by adding '-amine' as a suffix to the name of the parent compound. In substitutive nomenclature, the prefix 'amino-' is placed before the name of the parent compound to denote the fuctional group in high molecular amines. Synthetic amines are made mostly by reaction of alcohols with ammonia, catalyzed by metals( nickel or copper) or metal oxide at high temperature. Many methods have been devised for the synthesis of the amines reacting ammonia with an alkyl halide and neutralizing the resulting alkyl ammonium salt with an alkali, e.g., sodium hydroxide. This procedure yields a mixture of primary, secondary, and tertiary amines that is easily separated into its three components by fractional distillation boiling methyl isocyanate with caustic potash, heating the alkyl iodides with ammonia reduction of nitriles with alcohol and sodium heating the esters of nitric acid with alcoholic ammonia reducing on nitro-paraffms action of zinc and hydrochloric acid on aldehyde ammonias reduction of the phenylhydrazones and oximes of aldehydes and ketones with sodium amalgam in the presence of alcohol and sodium acetate action of dilute hydrochloric acid on the isonitriles heating the mustard oils with a mineral acid, by the hydrolysis of the alkyl phthalimides. Primary amines contain the functional group -NH2 (called amino group) and are converted into secondary and tertiary amines if heated with alkyl or aryl iodides. Primary amines form various oxidation products violently with concentrated nitric acid. If the amines are acetylated, they form nitro derivatives with concentrated nitric acid. Primary amines form diazonium salts with nitrous acid in cold solution in the presence of excess of mineral acid. Or a diazoamine is obtained in absence of excess of acid. Other ractions forming condensation products with aldehydes forming anilides forming alkyl thioureas yielding isonitriles with alcoholic potash and chloroform. Aliphatic amines which have the lowest carbon content are water-soluble gases or liquids of low boiling point also readily soluble in water in case of the next low carbon content. But aliphatic amines which have the high carbon content are odourless solids of high boiling point and are insoluble in water. They are all bases and easily forms salts with the mineral acids and solid salts with the halogenoalkanes. Amine Salts are crystalline substances that are readily soluble in water. Many insoluble alkaloids (e.g. quinine and atropine) are used medicinally in the form of soluble salts. If alkali (sodium hydroxide) is added to solutions of such salts the free amine is liberated. Hexamethylenediamine used in the manufacture of nylon-6,6 is prepared by catalytic addition of hydrogen to nitriles. Aromatic amines also exist, such as phenylamine, which are important for the production of diazonium salts. They dissociate in water (some very weakly). Aromatic amines are much weaker bases than the aliphatics. One of the most important aromatic amines is aniline pale brown liquid boiling at 184 C, melting at -6 C. Aniline is obtained commercially from chlorobenzene by heating with ammonia in the presence of copper catalyst or from a product of coal tar (nitrobenzene) through the reduction reaction. Aniline is the starting material in the dye manufacturing industry and as in the manufacture of others. Aniline is converted into sulfanilic acid which is the parent compound of the sulfa drugs. Aniline is also important in the manufacture of rubber-processing chemicals, antioxidants and varnishes. Amines take part in many kinds of chemical reactions and offer many applications include in agrochemicals, dyestuffs (the best known being aniline), pharmaceuticals, and corrosion inhibitors.
|
